
Reilly, James M. The Albumen & Salted Paper Book: The history and practice of photographic printing, 1840-1895. Light Impressions Corporation. Rochester, 1980.
| [Previous] Chapter 2 | Title Page | [Next] Chapter 4 |
| Table of Contents | Search this book |
A print on plain paper may be dead, foggy, inky, sunk in the paper, &c., &c., but in its most unhappy state it does not look vulgar; there is always a certain sentiment about it, even in its worst phase of failure.
--Thomas Sutton, 18561
Although others before him had experimented with the light-sensitive properties of silver chloride, it was the Englishman William Henry Fox Talbot who prepared the first successful silver chloride photographic paper. This discovery came in the years 1834-1835,2 and laid the foundation for all the subsequent printing-out papers that were so extensively used in the 19th and early 20th centuries. The details of Talbot's method were not revealed until 1839,3 after news of Daguerre's success had spread around the world. Talbot's "photogenic drawing" paper was prepared in two steps: first, fine quality writing paper was immersed in a weak salt solution and dried. The second step involved making the paper light sensitive by brushing on a strong solution of silver nitrate. The sensitized paper darkened when placed in sunlight, and was "stabilized" against further darkening (at low light levels) by washing the paper in a strong salt solution.
At the suggestion of Herschel, Talbot changed to sodium thiosulfate, or "hypo" as the fixing material, and this made the prints completely stable to light and left them with white highlights, instead of the pale lilac highlights characteristic of fixation in strong saline solutions. With the adoption in 1839 of "hypo" as the preferred method of fixation, Talbot's "photogenic drawing" paper had evolved into what is now known as "plain salted paper", the positive printing material that was to become almost universally used during the decade 1840-1850. During that time plain salted paper was commonly referred to as "ordinary photographic paper", to distinguish it from various types of develop-out papers.
Talbot had tried to use his "photogenic drawing" paper in the camera to make negatives, but exposures were so long that he abandoned print-out negatives as soon as a practical alternative was available. He retained the print-out method for positives, however, because the results were more controllable and in his eyes more aesthetically pleasing. He thus set the pattern for the whole 19th century; development was regarded as a necessary evil for negatives, and as too troublesome and unpredictable for positives. Also, the greenish-black color of developed positives was widely reviled as "inartistic."
Although he himself employed only a plain salt solution as the first step in making the paper, Talbot's prints were reddish brown in color and reflected the presence of gelatin, used by the paper manufacturer as a sizing material. French photographers found that the papers they used did not produce the same results as English papers did, because starch sizing was used by the French paper-makers instead of gelatin. Until the late 1840's no organic binder materials were used in the salting solutions for salted papers, only chlorides--usually table salt or sea salt. Photographers were at first quite puzzled by the different results from English and French papers, but eventually two French scientists, Alphonse Davanne and Jules Girard, investigated and explained the phenomenon.4

Fig. 9. A facsimile of a photogenic drawing that appeared in The Magazine of Science for April 27, 1839.
Plain salted papers are those which have little or no organic binder material, and are thus matte-surfaced and of relatively lower maximum density. However, for even a simple salted paper, some organic material is necessary; it must either be present in the paper before the salting solution is applied, or it must be added to the salting solution itself. In practice, it is usually more convenient and satisfactory to add organic material to the salting solution. The sizing that already exists in papers is usually insufficient, and without additional organic binder material the resultant prints would appear too flat.
A salted paper which closely approximates the printing materials of the 1840's can be made as follows:
| Sodium chloride | 20 g |
| Gelatin | 2 g |
| Water | to make 1 liter |
Swell the gelatin in 250 ml of cold water, then heat the rest of the water and in it dissolve the sodium chloride. Mix the hot water and gelatin together, and when it has cooled to approximately 80°F, it is ready to use. The paper is salted by floating it on the salting solution for three minutes. This solution contains far less gelatin than modern gelatin emulsion papers; the amount of gelatin is only enough to slightly inhibit the sinking of the image into the paper fibers. Hence the choice of rawstock for this or any other plain salted paper that does not contain very much viscous binder is extremely important and has a great influence on the results. Porous rawstocks such as watercolor paper will yield very flat prints indeed; early photographers generally chose the smoothest rawstocks available to them, and for plain salted papers that are not made with viscous binders it is best to imitate their choice of a fairly smooth paper.
A variant on the above formula for plain salted paper is:
| Sodium chloride | 20 g |
| Sodium citrate | 20 g |
| Gelatin | 2 g |
| Water | to make 1 liter |
The addition of a neutral citrate will cause the prints to be more reddish in color and slightly more "brilliant." However, the porosity of the rawstock will still be the largest single factor in the resultant prints. Both of these simple salted papers will require the same sensitizing, toning, and fixing. Detailed descriptions of these operations will be found in later chapters.
With whatever paper stock has been chosen, examine the paper closely before salting it to see if there is a difference between sides--usually there is a front side and a back side, meaning that one side is smoother and has more sizing than the other. The front side is the smoother one, and that is the side that should be coated with the salting solution. If there is a watermark visible, then the front is facing up when the watermark is readable. Once the paper has been salted and dried, there will be no way to tell which side has been treated, so it is necessary to mark the back of each sheet in pencil--to keep track of which side has been salted.
Floating is the preferred method for coating most salted papers, because it eliminates the possibility of patches of uneven density which might be caused if both sides are "salted." These patches of uneven density result when some of the silver solution is drawn to the back of the sheet during sensitization, thus locally sensitizing some areas on the back and causing a blotchy appearance on the face of the print. With some rawstocks, however, the simple gelatin salting solutions given above will provide good results when the paper is immersed and not floated. Only experience will determine which rawstocks can be successfully salted with the immersion technique.
The technique of floating is not difficult, and it actually becomes quite efficient and routine if the proper conditions are maintained. If the temperature of the paper itself and the salting solution is the same, the floating of the paper is made considerably easier. If the salting solution is warmer than the room temperature--which is also the paper temperature--then the sheets of paper will curl ferociously when they are placed upon the surface of the solution. In general, relatively thin papers are preferable because they are easier to handle, especially after they have been coated with binder materials and must be floated a second time on the sensitizing bath. Paper becomes stiffer when dry, and so a certain amount of moisture in the storage environment of the paper, especially just prior to floating, makes the sheets easier to handle.
Fig. 10. Salting paper by immersion.
Curling is caused by the swelling of the paper fibers on the bottom of the sheet, so if curling does occur it can be remedied by dampening the fibers on the top side of the sheet. Old manuals recommend breathing on the sheets to unbend them after they have curled, but this is impractical for all but the mildest curling.
After a sheet has rested on the surface of the solution for awhile, it will uncurl itself and gradually lie flat. But if the curling has been too severe, the sheet may be already ruined from the solution running on to the back. Sometimes holding the edges of the sheet down on the surface of the solution until the sheet uncurls itself is a workable method, but with large sheets this may take two people. With whatever method is chosen, it is always best to try to have conditions of moisture and temperature as favorable as possible before attempting to float. One way to control curling is by dampening the back of the sheet with a fine mist of water after it is placed on the solution. The inexpensive plastic atomizers sold for various garden uses will work well, but large droplets of water must not be allowed to collect on the back of the sheet while it is floating, and only a minimum of dampening should be used. An alternative approach, which works well for some people, is to fold back about ½ inch of paper on all four sides of the sheet, thus making a kind of "boat". The creased edges resist the tendency to curl and the folded flaps help prevent the solution from reaching the back of the sheet.
Many old manuals advise that paper be floated by handling it by two diagonal corners, bending the sheet and placing the center of the bent sheet on the surface of the solution. It is then gently lowered until the corners touch down and the sheet floats on the surface. This technique nearly always results in a line of bubbles becoming trapped under the sheet in a diagonal line from the two corners not held in the hands.
A better way is to first fold over about ¼ inch of paper in two opposite corners of the sheet, so that these two bent corners act as little handles to give better control of the sheet. Grasping the paper by these two bent corners, bow the sheet and place one corner of it on the solution. Gently lower the other hand until the whole sheet rests on the solution. The advantage of this approach is that the bubbles that may have formed when the sheet first touched down on the surface are forced out to the edges by the rolling motion from one corner to the other, instead of from the middle to the two corners at once as the old manuals suggest.
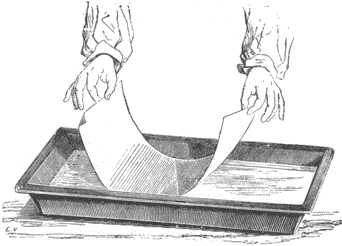
Fig. 11. Salting paper by floating it on the salting solution.
If any bubbles do appear, lift the sheet from one corner and if they do not break of their own accord, pierce them with a toothpick and replace the sheet on the surface. Bubbles may usually be seen through the paper itself, but if the stock is too thick, then the sheets will have to be lifted from the corners to check for bubbles. Time the operation of floating from the point at which all bubbles have been broken and the sheet rests evenly upon the surface.
When the sheets have floated for the required length of time, carefully lift one corner and slowly raise the sheet from the solution. The sheets will be coated more evenly if they are slowly peeled from the surface. By slowly lifting them there will be less dripping of liquid back into the tray of solution, and fewer bubbles will be formed in the tray to interfere with the next sheet to be floated. Always check the surface of the solution for dirt or bubbles before attempting to float a sheet of paper.
The sheets should be hung to dry by the two corners of the long edge, so that the downward runoff has the least distance to travel. This minimizes the unevenness of Coating which always occurs because the solution runs downward toward the floor, forming a slightly thicker coating on the lower half of the paper. If the sheets curl during drying, clothespins may be clipped to the bottom edge of the sheets to weight them down. Heat may be used to accelerate drying, but with plain salted papers there is no advantage gained except faster drying.
When the sheets are dry, they should be taken down and pressed flat under some weights. This makes them more supple and easier to handle in the sensitizing step.
The paper will keep indefinitely in this salted condition, if stored in a cool and dry location. Of course all salting operations may be conducted in white light, since the paper is not light sensitive until it has been sensitized on the silver nitrate solution.
Paper made according to the above two formulae should be sensitized by floating for 3 minutes on a 12% silver nitrate solution. After the paper is sensitized, however, it will keep for only one or two days, depending on the temperature and humidity of the storage environment.
Each type of silver printing paper has a characteristic gradation and contrast. This means that only a certain type of negative will give the optimum results with each kind of salted paper. For example, the plain salted papers given above are very soft-working papers and require negatives of far greater density range than any modern develop-out paper. The reasons for this have to do with the basic nature of printing-out papers and the smoothness or porosity of the paper stock. A detailed explanation of the tone reproduction characteristics of albumen and salted papers will be given in Chapter 7, but briefly stated, the more matte-surfaced a paper is, the lower the contrast of that material is likely to be. Albumen paper, therefore, requires a less contrasty negative than plain salted papers.
Techniques for increasing the contrast of printing-out papers do exist, and they can be especially useful in printing negatives with a less than optimum density range. In the 1840's the paper negative processes then in use were well suited to salted paper printing, since they often produced "harsh" negatives. For modern negatives that have been made for printing processes with greater inherent contrast, it is helpful to have a means by which such negatives may be printed on salted paper. Controlling the intensity and color of the exposing light is one convenient way to control contrast, and this is discussed more fully in Chapter 7. The chloride content of the "salting" solution also has an effect on contrast.' Large increases of contrast are possible only with the use of chromates, and this technique does allow otherwise unprintable negatives to produce good prints. Nevertheless, the judgment of years of experience by many different writers on silver printing is that the best salted paper prints are always made in sunlight with optimum negatives, and without resorting to contrast enhancing additions to the salting solution.
The addition of chromium salts to the salting solution is the basic method of contrast enhancement. This technique is presented here only as an alternative for use when a print is desired from a too "thin" negative; it has no place in regular practice, since the finest results may only be obtained by using the "pure" processes with optimum negatives.
The discovery that chromium salts led to higher contrast was made by Ferdinand Hrdliczka in the mid-1890's--a time when printing-out papers were gradually losing ground to the developing-out papers.6 For most of the 19th century, photographers depended mainly on control of the negative density for contrast control in the print; that is why so many intensifier formulae are found in 19th-century photographic manuals. Chromated papers were introduced to the market in the late 1890's, but their sales amounted to only a small portion of the total sales of printing-out papers.
Potassium bichromate is the chromium salt most often employed for the purpose of increasing contrast. A convenient way to use it is to make a 5% stock solution with distilled water. CAUTION! Potassium bichromate is a toxic substance and should be handled with care. Some individuals are extremely sensitive to it, and all contact with the skin should be avoided. Wear eye protection and gloves, and avoid inhalation of dust by wearing a disposable surgical mask when mixing the bichromate solution.
Always work in a well ventilated area.
The bichromate stock solution is added to the salting solution in varying amounts depending on the degree of contrast enhancement desired. The effect of bichromate additions is mainly on the highlight areas of the image. A sample salting solution containing bichromate is:
| Sodium chloride | 25 g |
| 5% solution Potassium bichromate | 5 ml |
| Gelatin | 2 g |
| Water | to make 1 liter |
Sensitize for 3 minutes on a 10% silver bath which also contains 5% citric acid.
It is best to keep bichromate additions to a minimum. The stability and predictability of papers declines with increasing amounts of bichromate. The practical limits of contrast control with this technique will have to be established through experience. In no case should the dry weight of bichromate exceed 1% of the salting solution. The disadvantages of bichromate additions include the toxicity of the substance, the fact that papers must be salted and stored in yellow or diffused white light because of the light sensitivity of potassium bichromate itself, and the necessity of avoiding any contact of the salting solution with metals, since these react unfavorably with bichromates. Also, the addition of bichromates tends to produce a browner image color than would otherwise be obtained. Bichromate additions will serve the same contrast-enhancing function in almost any kind of printing-out paper, and they are employed in approximately the same quantity with other papers as with the plain salted paper formula given above.
Arrowroot paper is a form of salted paper in which the binder material is a paste made from boiled arrowroot starch. The surface qualities of arrowroot paper range from very matte to a dull gloss, depending on the amount of starch applied to the paper and the smoothness of the underlying rawstock. Arrowroot prints can be considerably more brilliant and richer-looking than plain salted papers; they have a longer density range and preserve more delicate detail.
The first starch papers were prepared by DeBrébisson in 18547 using tapioca instead of arrowroot, but eventually arrowroot emerged as the most suitable and widely used starch for the purpose. Starch papers became very popular and almost completely displaced plain salted papers for matte-surfaced prints. In 1854, the popularity of albumen paper was increasing as well, but a significant number of photographers still preferred a matte surface on their prints. At that time, albumen paper was very new and had its share of problems--it was harder to tone and fix than other kinds of paper, and it was sometimes difficult to coat evenly and the newly discovered starch papers represented a great advance over the older plain salted papers. The middle 1850's saw the rise of a new kind of business enterprise offering salted and sized photographic papers to the public as an article of trade--and many of the new companies sold both albumen and starch papers.
However, significant improvements in albumen paper were not long in coming, and during the last half of the 1850's albumen paper ascended to the unchallenged dominance of the photographic paper market that it was to keep for the next 35 years. Although diminished in popularity after 1860, starch papers continued to be used by a small number of photographers, and arrowroot paper remained an article of commerce well into the 20th century. Unfortunately, starch papers appeared at a time when the public taste was very much on the side of glossy papers, which were a novelty at the time and better suited to the small but immensely popular cartes de visite size portraits. In a typical full-length pose, the head of a person in a carte de visite might be less than ½ inch high, so maximum detail was necessary in the print to make such a tiny image successful.
After 30 years, public taste began to change again, and glossy papers fell out of favor. In the 1880's the first awakening of a renewed interest in matte papers was felt, led by photographers whose main interest in photography was aesthetic, not commercial. By 1900, the public at large had come to regard matte papers as more "artistic," and although there were a great number of printing methods to satisfy the demand, arrowroot papers enjoyed a modest revival along with plain salted papers. The application of platinum toning methods allowed a whole range of brown tones to be produced, and this helped increase public acceptance of arrowroot papers. The poor economics of professional portraiture with expensive and time-consuming printing-out papers eventually drove the last arrowroot and plain salted papers off the market in the years following World War I.
Arrowroot prints can be made on almost any sort of paper surface, but a fairly smooth surfaced paper is generally preferred. Highly calendered "plate" finish papers present a little more difficulty in obtaining an even coating. Arrowroot pastes tend to be absorbed into the paper fibers--in fact, they must be absorbed to a certain extent to adhere--and so porous papers will require a binder that contains a higher percentage of starch.
The salting solution is prepared as follows: rub 35 g of arrowroot into a creamy paste with a little cold water. This is best done in a mortar and pestle. Add enough cold water to make a fairly runny "cream" with no lumps. This "cream" will tend to separate into its components, so it must be stirred or rubbed up to the moment of use. Separately dissolve 35 g of sodium chloride and 3 g of citric acid in 950 ml of water. This solution should be brought to a boil in a porcelain container, and the arrowroot cream added in small amounts with constant stirring using a glass rod or wooden spoon. Allow the mixture to gently boil for a few minutes and then remove from the heat. When the mixture has cooled it is ready to use. Remove the skin that forms on the surface of the cooled liquid; this is the residue of the hulls of the burst grains of starch. Tapioca or rice starch may also be used in the same manner as arrowroot.
The formula for arrowroot paper is essentially the same as for plain salted paper except that it contains citric acid. Starch is not an "active" organic substance and so has no effect on the reduction of silver chloride. Hence if the citric acid which is "active" were not present, the prints would be gray and flat, and hallmarks of a pure chloride image. In the presence of both citric acid and starch the prints take on a warm purple color after exposure, and, in the absence of toning, change to a yellowish-brown after fixation.
There are many approaches to applying the starch salting solution to the paper. Always mark the back of the sheet in pencil before applying the salting solution. Salting solutions that contain 2% arrowroot or less are fluid enough to allow the paper to be floated on the starch solution. This is a little more difficult than floating paper on albumen or thin gelatin solutions, and will not provide as much binder on the surface of the paper as other methods. Heavier coatings of starch may be obtained by immersing the sheets in the salting solution and drawing them out over a glass rod, or between two glass rods held tightly together.
The method most recommended in old manuals is first to pin the sheet of paper to a flat board, and use a thin flat brush to apply a 3-4% starch paste. The salting solution is lightly applied by brushing first in one direction, and then distributed with perpendicular cross-strokes. The paste is allowed to sink in for a minute or two, then a round dry brush is used to even out the coating and remove excess paste until a uniform matte surface is obtained. The difficult part is to keep the coating as even as possible at every step of the operation. If the paper is textured, care must be taken to insure that the paste is uniformly brushed into all the crevices. Still another approach is to apply the paste with a sponge, wait 1 or 2 minutes, and scrape off the excess with a squeegee, first in one direction, then the other. The paper is hung to dry after it has been coated with the salting paste. Only experience will determine which is the best approach to coating the paper for any given paper stock or individual.
Arrowroot paper should be sensitized by floating on a 12% silver nitrate solution that also contains 4-5% citric acid. The length of time for floating starch papers on the silver bath is generally shorter than with other papers. If the coating of arrowroot is not a heavy one, then ½ minute may be sufficient. For heavily coated papers, 1 ½ minutes at most may be required. Too long floating on the silver bath results in gray and flat prints, especially with porous papers. The silver nitrate solution may also be applied with a wide brush. Citric acid additions to the silver bath will produce paper that will keep several weeks after sensitization. The toning of arrowroot papers may be done with any of the toning formulae given in chapter 8.
The individual operations in the processing sequence of salted papers--toning, fixing, washing, and drying--are dealt with in depth in separate chapters. For the sake of clarity, a brief overview of all the processing steps for a typical albumen or salted paper print is as follows:
In order for the papers to have the necessary sensitivity, they must contain an excess of silver nitrate (see chapter 1). The initial wash--usually about 10 minutes in running water--is the first step in processing and it serves to remove this excess silver nitrate. If it were not removed at this stage, the silver nitrate would retard or completely prevent any toning from taking place, and if it were still present when the print is fixed, black stains would be the result. In large-scale printing operations the first wash water is carefully saved, and from it the majority of the silver used to sensitize the paper is recovered
The toning process is the largest single factor in determining the final color of the print. It is performed before the fixing step--although it may be done after fixing with equal success--because the toning process forms silver chloride as a by-product, and this would therefore resensitize the print if it had already been fixed. The toning step is performed in weak white light if the prints are to be toned by inspection and not "by rule." Otherwise all the processing steps up to and including the fixing are performed in yellow light.
A short wash--3 to 5 minutes in running water--is given before the prints are transferred to the fixing solution. Interaction between the fixer and toner may cause changes in the fixing solution that would damage the color and permanence of the prints.

Fig. 12. A processing line for albumen prints. Trays B & C are for the initial wash; D is the toning tray, and E is the fixer tray. F and G are for washing the prints.
The purpose of fixing is to remove the unreduced silver chloride and other light-sensitive substances such as silver citrate, etc., which may be present. A fixer composed of an alkaline 15% solution of sodium thiosulfate solution is useful for all albumen and salted papers. The time of fixing should be 8 to 10 minutes. For maximum permanence, two fixing baths are used. The prints should be fixed for 4 minutes in each bath, and drained for at least 5 seconds before being placed in the second fixing bath. A fresh batch of fixer should be made for each printing session; the extra silver present in salted papers quickly exhausts fixing baths, and even unused fixer solutions break down very rapidly.
The prints should be given a short (2 to 4 minutes) wash in running water before treatment with a hypo clearing agent. This wash removes a large portion of the hypo and avoids overloading the mechanism of hypo removal.
The use of a hypo clearing agent is recommended to aid in the more complete removal of silver-thiosulfate complexes which may cause damage to the print. A 3-to 4-minute treatment in 1% sodium sulfite solution is required.
Depending on the thickness of the rawstock, the prints should receive at least a 30-minute wash in an effective print washer. Heavier weight papers may require longer washing.
Prints may be dried in a number of ways. A good approach is to gently blot them on high quality photographic blotters and air dry them face up on fiberglass screens.
| [Previous] Chapter 2 | Title Page | [Next] Chapter 4 |
| Table of Contents | Search this book |