ANALYSIS OF LAMINATED DOCUMENTS USING SOLID-PHASE MICROEXTRACTIONMark Ormsby
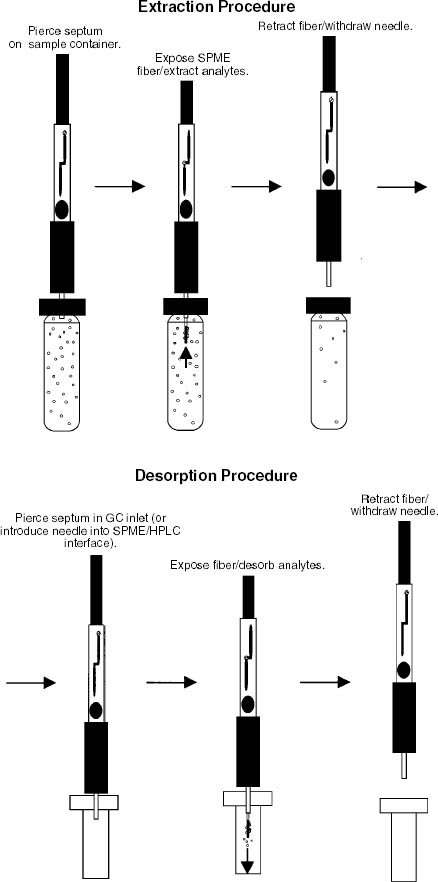
2 BACKGROUND2.1 SOLID-PHASE MICROEXTRACTIONOne of the principal advantages of SPME is its simplicity (Vas and Vekey 2004). In one solvent-free step, the SPME fiber collects and concentrates the sample, and the fiber is then used to transfer the sample to the instrument for analysis. Other sampling procedures may require several time-consuming steps using solvents and expensive equipment while increasing the potential for sample loss and contamination. In many cases, the detection limits with SPME are also lower than with other methods. The procedure for collecting the sample is shown in the top row of figure 1 (Supelco 1999). Similar to a syringe, a SPME fiber has a 1 cm length of fused silica attached to a stainless steel plunger. The tip is coated with a polymer and is shielded inside a hollow needle. When the plunger is depressed, the fiber extends, the polymer is exposed, and the sample is collected onto it by absorption or adsorption, depending on the type of coating. After a suitable exposure time (a few seconds to a few hours or even weeks, depending on the application), the fiber is retracted. The bottom row of figure 1 illustrates the process for transferring the sample for analysis. The fiber is inserted into the GC-MS, and the plunger is depressed to expose the polymer. The heated injector port drives off the collected compounds, which then flow into the instrument for qualitative or quantitative analysis. Since the fiber has been “cleaned” by heating in the injector, it is ready to be reused.
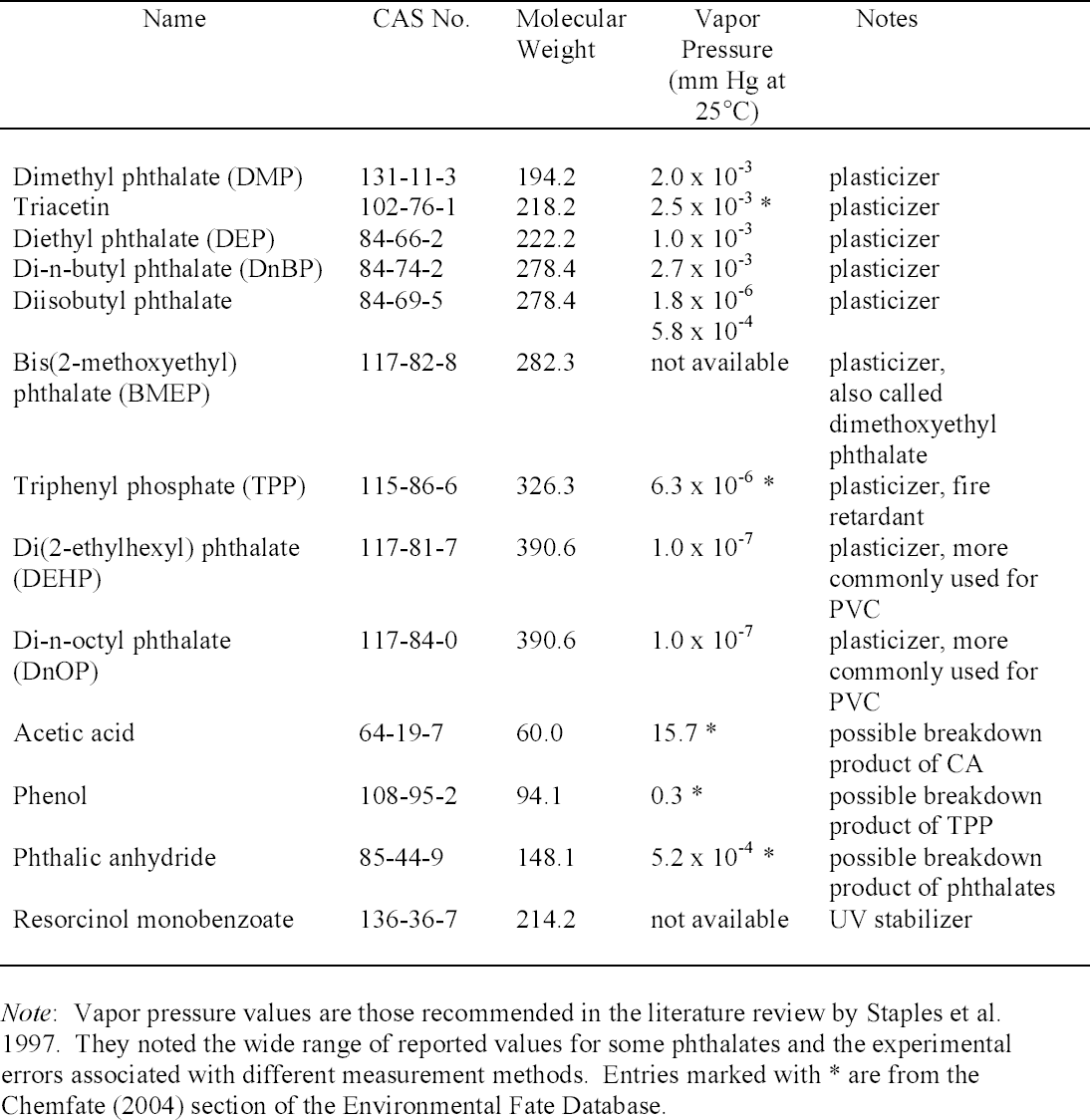
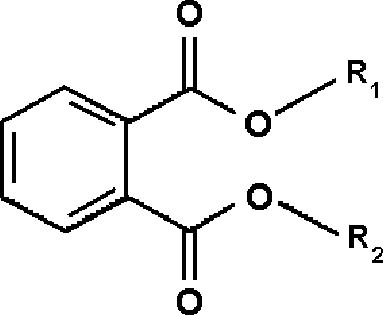
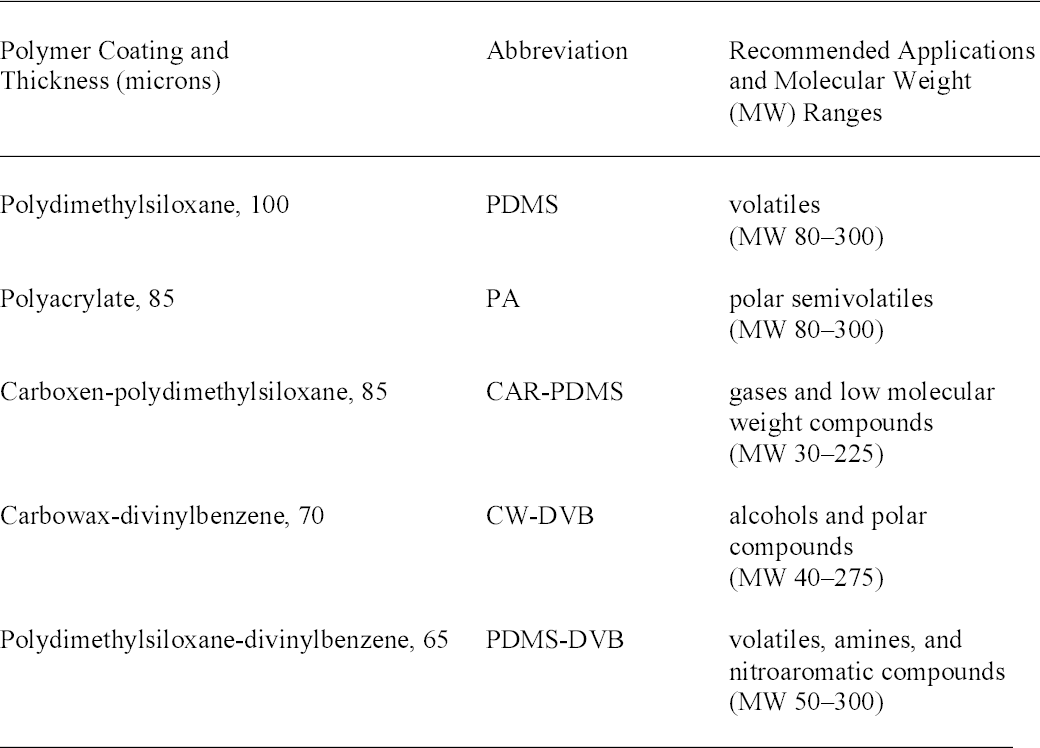
With direct immersion (DI-SPME) sampling, the fiber is inserted into a liquid. In headspace (HSSPME) sampling, the fiber collects compounds from the headspace above the sample. HS-SPME reduces interferences from the sample matrix and is often used for analysis of soils, wastewater, blood, and other complicated matrices (Vas and Vekey 2004). The amount of sample collected on the SPME fiber depends on a number of factors (Supelco 2001). As with GC columns, a fiber coated with a polar polymer is more sensitive to polar compounds, and 2.2 ADDITIVES IN LAMINATION FILMSPlasticizers are compounds that dissolve in and modify the properties of a polymer but are not chemically bound to it (Williams 1993). Low molecular weight phthalates (fig. 2) are commonly used to plasticize CA (Stannett 1950) as well as other polymers. They have become widespread environmental contaminants because they readily leach or volatilize from plastic products during use or after disposal (Staples et al. 1997). Table 1 lists phthalates and other additives commonly found in
In addition to plasticizers, the CA film may contain a number of other additives. The NBS specification (Wilson and Forshee 1959b) for archival laminating film recommended a minimum of 0.5% by weight of an acid acceptor such as magnesium acetate, as well as an antioxidant. A minimum of 1% by weight of an ultraviolet absorber was also recommended. To effectively use SPME to study the variety of plasticizers and other additives in lamination films, it is necessary to determine which fibers are most sensitive for these compounds. A set of CA films was studied systematically by both HS-SPME and DISPME. These samples were prepared by NBS as part of their research programs in the 1950s and are now in the files of the NARA Research and Testing Laboratory. These results were applied to SPME analysis of actual laminated documents.
|