ALVAR AND BUTVAR: THE USE OF POLYVINYL ACETAL RESINS FOR THE TREATMENT OF THE WOODEN ARTIFACTS FROM GORDION, TURKEYKRYSIA E. SPIRYDOWICZ, ELIZABETH SIMPSON, ROBERT A. BLANCHETTE, ARNO P. SCHNIEWIND, MAURAY K. TOUTLOFF, & ALISON MURRAY
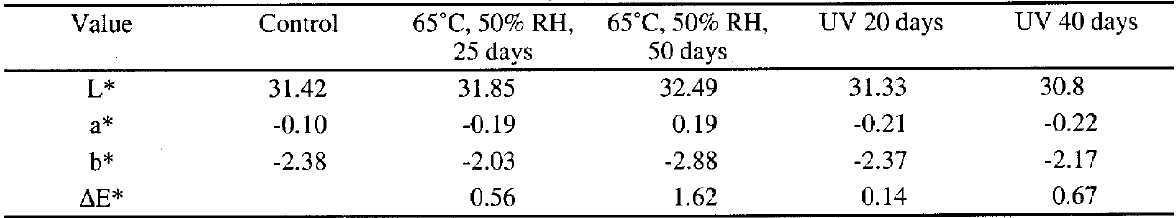
7 EXPERIMENTAL—EXPOSURE TO INTENSE ENVIRONMENTAL CONDITIONSThe purpose of this study conducted at Queen's University, Kingston, was to give preliminary information about the aging characteristics of Butvar B-98 resin (Toutloff 1999). The material properties of Butvar B-98 can be found in the product literature (Solutia Inc. 1999); for example, the glass transition temperature (Tg) is stated as 72-78�C. Both chemical and physical properties were selected for analysis after the samples were exposed to intense environmental conditions. These conditions included high temperature at constant relative humidity, as the product literature implies that Butvar B-98 may cross-link upon exposure to heat. Long-wave ultraviolet light was also chosen as an experimental parameter because it would introduce energy into the system, which would increase the probability of chemical reactions that might lead to cross-linking between and within the polymer chains. Infrared spectroscopy was used to examine the samples for evidence of cross-linking after exposure. The physical properties of color, hardness, and solubility were measured to detect any differences between the exposed samples and the control group, due to chemical changes. 7.1 MATERIALSixty samples were produced by casting layers of a 15% w/v solution of Butvar B-98 in 60:40 ethanol and toluene with an average thickness of 0.03 mm onto glass microscope slides. The samples were allowed to dry for seven days to ensure complete solvent loss. 7.2 CONDITIONSGroups containing six samples each were exposed to different environmental conditions for specific amounts of time: (1) a temperature of 65�C and a relative humidity (RH) of 50% for 25 days; (2) a temperature of 65�C and 50% RH for 50 days; (3) long-wave ultraviolet radiation for 20 days (20-25�C and 50-60% RH); (4) long-wave ultraviolet radiation for 40 days (20-25�C and 50-60% RH); and (5) dark storage (20-25�C and 50-60% RH) for the control. A Tenny model TH65 Versa Tenn 65 cu. ft. temperature–humidity environmental chamber was used to obtain an environment of 65�C and 50% RH. The long-wave radiation was emitted from a 365 nm long-wave ultraviolet bulb. 7.3 METHODSInfrared spectroscopy was performed using a Perkin-Elmer Model 983G double-beam ratio recording infrared spectrophotometer. The spectra were recorded at a nominal resolution of 4 cm-1.A KBr pellet was made using 1-2 mg of sample scraped from the Butvar B-98 film. A MacBeth Color-1 Colorimeter measured L* (lightness), a* (green-red) and b* (blue-yellow), using the standard illuminant D (average daylight, 6500�K),
Hardness was measured by indentation with a Shore Durometer A Type D, ASTM Designation D2240, which uses a pointed steel pin. The instrument had a constant load-operating stand and a deadweight of approximately 822 g. Hardness measurements were taken from three different areas on three samples for each condition, for a total of nine measurements. Two methods were used to test for solubility. The first involved placing samples in 60:40 ethanol and toluene solvent mixtures. As all samples dissolved immediately, a second method was used. A swab that was saturated with the same solvent mixture was passed over the film until the film completely dissolved. The number of passes was counted. The experiment was carried out by one person, in as consistent a manner as possible. The measurement was made twice for each condition. 7.4 RESULTS AND DISCUSSIONThe IR spectra showed little difference before and after exposure to the intense environments, indicating that there were no substantial chemical changes. No new peaks appeared after exposure that would indicate the formation of new bonds due to cross-linking. A minor change occurred around 1106 cm-1 and 1138 cm-1 in the samples exposed to high heat and humidity for 50 days, most probably indicating absorption of -OH groups due to water from the humid conditions. Color change was not apparent to the naked eye. The measurements showed that there was minimal change, which is consistent with the literature. The average δE* was over 1 only for the samples exposed to 65�C and 50% RH for 50 days (δE* = 1.62) (table 2). All samples exposed to ultraviolet light had δE* less than 1. The results from the hardness tests show that Butvar B-98 became only slightly harder after exposure to intense environmental conditions. Most of the samples showed an increase in hardness of 1 unit (from 15 units), which is very small. There were only two exceptions where one of the three areas on the samples had an increase of 2 units. These samples had been exposed to intense environmental conditions for the longer times (50 days at 65�C and 50% RH, and 40 days of ultraviolet radiation). The first solubility test demonstrated that the films were extremely soluble in the solvent mixture and could be considered reversible. The second method using solvent-saturated cotton swabs showed that samples exposed to 65�C and 50% RH for 50 days required 15 passes and were therefore the least soluble; samples exposed to ultraviolet radiation for 20 days required 13 passes; samples exposed to ultraviolet radiation for 40 days required 12 passes; samples exposed to 65�C and 50% RH for 25 days required 8 passes; and the control required 9 passes. 7.5 CONCLUSIONThe results from the infrared analysis taken in conjunction with the results of the physical tests indicate that the Butvar B-98 resin film remained fairly stable under the intense environmental conditions of the experiment. The group that showed the most change in all cases was the set exposed to a temperature of 65�C and 50% RH for 50 days. These results concur with the product literature, which |