CELLULOID OBJECTS: THEIR CHEMISTRY AND PRESERVATIONJULIE A. REILLY
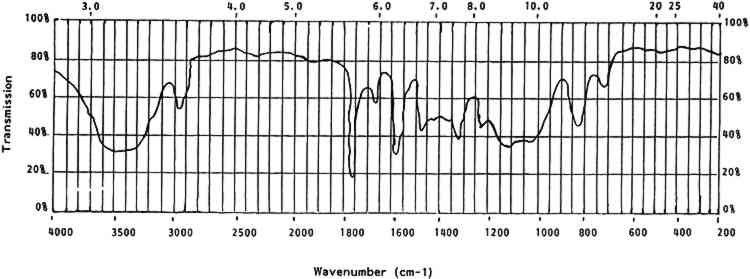
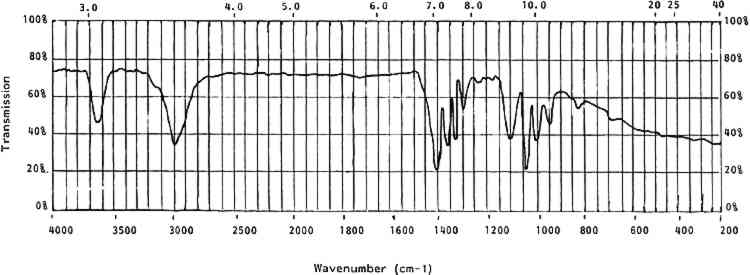
4 CELLULOID IN THE COLLECTION4.1 IDENTIFICATIONTHE FIRST step in dealing with celluloid objects in a museum setting is identification. This is rarely possible by visual or tactile means; chemical or analytic identification is necessary. This section deals with the identifying properties and characteristics of celluloid. 4.1.1 General CharacteristicsCelluloid is a rigid thermoplastic made by compression or blow molding, casting, or machining. It softens in moderate heat. When gently rubbed with a soft cloth, celluloid will give off the odor of camphor. It burns rapidly and continually with a yellow flame when ignited. It may explode or give off the odor of camphor while burning (Saunders 1966, 18). Celluloid contains nitrogen that can be detected by a variety of techniques (Saunders 1966, 21). The presence of nitrogen can be an indicator of celluloid, but it can also indicate other nitrogen-containing polymers. However, the presence of nitrogen can be used to distinguish celluloid from other cellulosic derivatives. 4.1.2 Optical CharacteristicsCellulose has an optically biaxial and positive unit cell and is highly birefringent. When nitrated the fibers become less birefringent and the optical activity changes from biaxial positive to uniaxial to biaxial negative with increasing nitrogen content. The refractive index of cellulose nitrate is lower than that of cellulose and drops even lower with increasing nitrogen content. The range is from 1.49 to 1.51 (Miles 1955, 210–11). Isolation of cellulose nitrate from celluloid for optical identification is problematic in that when cellulose nitrate is gelled by camphor it loses its original structure and, hence, many of its original optical characteristics. As noted earlier, under low magnification celluloid can be indistinguishable from natural materials. 4.1.3 SolubilityCelluloid is soluble in two types of solvents. The first type includes acetone, other ketones, and alkyl acetates. These solvents do not cause any change in the crystalline structure of the cellulose nitrate molecule. The second type of solvent—methyl nitrate, alkyl nitrate, and water—does change the crystalline structure of cellulose nitrate molecules (Petitpas and Mathieu 1946, 525). The solubility of celluloid is complex in that it is a two-component system—camphor and gelled cellulose nitrate (table 5). Some solvents, such as ethanol, primarily affect the camphor portion while others affect both portions (acetone) and some affect only the cellulose nitrate (water). The solubilities of celluloid can differentiate it from natural materials. For example, celluloid is soluble in acetone while ivory is not. The method used by the author to differentiate celluloid from ivory involves taking a very small scraping from the object with a small scalpel blade under low magnification. The sample, visible under microscope, is left on the edge of the scalpel blade and a single drop of acetone is placed on the sample. Ivory flecks do not dissolve and are swirled around in the currents of the acetone drop. These flecks appear to sparkle as they reflect light. Celluloid dissolves instantly, leaving an apparently pure drop of acetone. TABLE 5 Solubility of Celluloid 4.1.4 StainsCelluloid can also be identified by several stain reactions. These reactions can be affected by the presence of certain common chemicals, so it is best to use as many different identification techniques as possible (table 6). TABLE 6 Stain Reactions for Identification 4.1.5 Infrared SpectroscopyOne definitive test for celluloid and cellulose nitrate is its characteristic infrared spectrum. Samples prepared in KBr pellets or as a thin film will provide a clear-cut set of peaks. Figure 3 shows a generalized spectra of aged celluloid and camphor. Table 7 lists the wavenumbers of cellulose nitrate.
TABLE 7 Cellulose Nitrate Infrared Spectrum All solubility, stain, and infrared tests given here have been tested by the author unless otherwise noted. Techniques that require unusual equipment, great exactitude, or a significant amount of time have not been included. 4.2 ISOLATION AND DOCUMENTATIONOnce an object has been identified as celluloid, it should be isolated from other materials so that its deleterious breakdown products will not harm other objects. After isolation, a condition report is essential. This documentation will sometimes be the only source of information to use in monitoring the condition of the objects over time. It is interesting to note that celluloid is often an unsuspected part of a collection until its effects on neighboring objects or materials are noticed. Certain measures that may slow inherent or environmental deterioration can be implemented only after celluloid objects are segregated. 4.3 CLEANING AND TREATMENTAfter examining the objects and recording their condition, note any film or droplets on their surfaces. These are generally acidic deterioration products and should be removed, as they will accelerate degradation of the celluloid. Little experimentation has been conducted to date on cleaning methods for celluloid. The deterioration products are usually readily soluble in water. However, water should be used sparingly as deteriorated celluloid can be water-soluble. Cleaning with a barely dampened swab can be effective but can also be abrasive. If water is used it should be removed immediately with a dry swab. Many celluloid objects have adhesive residues on them from pressure-sensitive tapes and glues. These adhesives have often combined chemically with the celluloid and can only be removed mechanically. Mechanical cleaning is very dangerous on celluloid because the object surface is often softer than the adhesive accretion. It is advisable to remove foreign accretions from celluloid, as they may accelerate local degradation, but only if extreme care and skill can be applied. After any deterioration products or accretions 4.4 STORAGE, EXHIBIT ENVIRONMENTThe instability of celluloid is both inherent and environmental. If the environment of a celluloid object, in storage or on exhibit, is adequately controlled, deterioration may be slowed but never entirely halted. The chemical and physical characteristics of celluloid deterioration indicate that an ideal environment for celluloid objects would include:
|