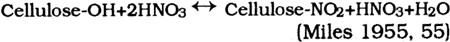CELLULOID OBJECTS: THEIR CHEMISTRY AND PRESERVATIONJULIE A. REILLY
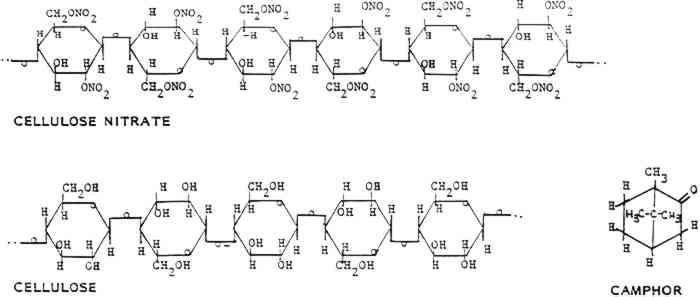
2 SYNTHESIS AND MANUFACTURE2.1 GENERALCELLULOSE NITRATE is created when the cellulose molecule is chemically modified by strong nitric acid, sulfuric acid, and water (fig. 1). The compound thus formed has been used to produce a wide variety of materials, including explosives, adhesives and cements, film bases, collodion, lacquers and coatings, and solid plastics (table 4). The primary differences among these various materials can be related to the degree of nitration of the cellulose nitrate molecule used in their manufacture. The level of nitration of a cellulose nitrate molecule is usually given as the percentage of nitrogen atoms per glucose
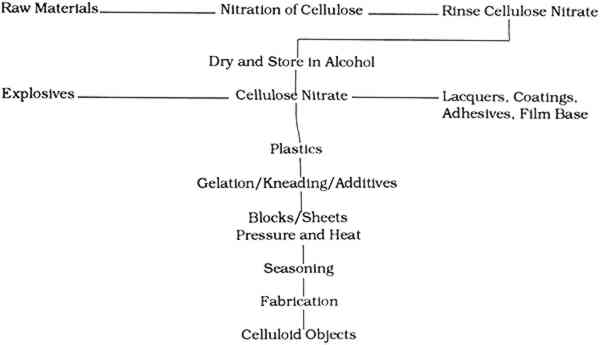
TABLE 4 Cellulose Nitrate-Based Products The synthesis and manufacture of celluloid plays a pivotal role in its long-term stability; in many cases, the production history of an object will determine its survival. The following discussion of the synthesis and manufacture of celluloid highlights those factors that directly or indirectly affect the long-term condition of objects in museum collections. 2.2 RAW MATERIALSThe raw materials required to synthesize celluloid include cellulose fibers, nitric acid, sulfuric acid, water, alcohol, camphor, colorants, and fillers (fig. 2). The cellulose fibers, acids, water, and alcohol are used to make cellulose nitrate, which is mixed with the camphor, colorants, and fillers to make celluloid.
2.2.1 CelluloseThe starting material, cellulose, can be obtained from a variety of sources, including cotton, ramie, flax, jute, and wood pulp (fig. 1). The main commercial source of cellulose for cellulose nitrate synthesis in the United States was cotton mill waste, including cloth scraps from clothing production and waste fibers from spinning plants. These wastes were processed by degreasing with carbon tetrachloride or benzene, scouring, bleaching, and washing to yield fairly clean short cotton fibers (Worden 1911, 1:38–39). Cotton mill waste was the most common source, but considering the economic or commercial nature of cellulose nitrate production, it is conceivable that any type and quality of cellulose may have been used to make cellulose nitrate. The inherent qualities of the original cellulose are partially retained by the finished celluloid. For this reason, one potential source of deterioration in celluloid comes from the instability of the cellulose molecule and the chemicals used to process the cellulose. 2.2.2 Acids, water, and alcoholThe nitric and sulfuric acids used were generally of high purity, although in more recent processes much of the acid mixture used is “waste acid” reclaimed from other processes (Worden 1911, 1:83–84). The water and alcohol used were generally of high quality, although lower purities of these liquids were also often used for economic reasons. Impurities in any of the above liquids may introduce centers of deterioration in finished celluloid. 2.2.3 CamphorAfter the cellulose nitrate is synthesized, it is mixed with camphor, a resin from the evergreen cinnamomum camphora that grows in Asia and Florida (fig. 1). The trunks, roots, and branches are steam distilled to yield the resin, which can be pressed and fractionated into several components, all consisting of variations of the general camphor molecule (C10H16O) (Brady and Clauser 1977, 126–28). The primary structure is 1,7,7-trimethylbicyclo(2,2,1)hepta-2-one (Selwitz 1988, 43). Camphor is a white crystalline substance that belongs to the terpenoid family. It is bicyclic and the natural form is dextrorotary. The specific gravity of camphor is 0.986 to 0.996. It sublimes (evaporates from the solid) at room temperature, melts at 176�C (350�F) and boils at 209�C (408�F) (Encyclopedia Americana 1983, 303). Camphor was first synthesized in 1859 but was not commonly made for industrial uses until after World War I (Kirk and Othmer 1982, 745–49). Synthetic camphor is produced by converting α-pinene from turpentine to camphene to isobornyl acetate to isoborneol to camphor. Synthetic camphor is chemically identical to natural camphor but is optically inactive (racemic) (Brady and Clauser 1977, 126–28; Kirk and Othmer 1982, 709–62). Several types of camphor substitutes have similar characteristics but different chemical structures. They include Borneo camphor (borneol) and bornyl chloride. Neither of these compounds was widely used but may have been mixed with camphor (Brady and Clauser 1977, 126–28). Other camphor substitutes developed by industry include Lindol (Celanese Corp.), which is tricresyl phosphate ortritolyl phosphate; triphenyl phosphate; dehydranone (Union Carbide); and cyclohexyl levulinate. None of these products offered better characteristics than camphor (for less cost). None was used to replace camphor entirely, although small amounts were often added to the celluloid mixture. Worden lists the characteristics that make camphor the best “latent solvent” for cellulose nitrate:
Worden writes that other “latent solvents” do not readily “gelatinize” cellulose nitrate, crystallize out, darken on exposure to light, soften, or produce other unwanted characteristics in cellulose nitrate plastics. 2.2.4 AdditivesColorants added to celluloid include dyes, pigments, and lakes. Other fillers and additives As the above discussion suggests, the raw materials used to create celluloid play an important role in the stability or instability of the finished celluloid. 2.3 Cellulose Nitrate SynthesisAs noted earlier, the first step in making celluloid is the synthesis of cellulose nitrate fibers (fig. 2). This synthesis is achieved by exposing cleaned cellulose fibers to an aqueous solution of nitric acid. Many of the two secondary hydroxyl groups and the one primary hydroxyl group on the glucose units
As the arrows indicate, this reaction is reversible in the presence of nitric acid and water. The nitrate groups oxidize the cellulose residues and produce nitrous oxides that catalyze further reactions. This reversibility of the nitration reaction indicates a source of potential deterioration. Early in the experimentation with this reaction, it was discovered that the presence of another acid in the nitration vat would produce more uniformly nitrated cellulose nitrate and help reduce the cost of reagents for the reaction. Water, acid salt mixes, other organic liquids, and several acids were added to reaction vats by researchers in an attempt to find the optimum cellulose nitrate. Most of these experiments were unsuccessful. Sulfuric and phosphoric acids were acceptable, but phosphoric acid proved to be too corrosive to the reaction vats at nitration temperatures and was only used for small batches of explosive-grade cellulose nitrate. Sulfuric acid proved to be the best additive. It slowed the nitration reaction, lending the manufacturers a degree of control (Miles 1955, 61–101). It produced more uniformly nitrated batches of cellulose nitrate and thus improved the quality of the cellulose nitrate produced. Once it had been established that a sulfuric/nitric acid mixture was best, much research was dedicated to deriving the optimum concentrations of each reagent in the mix. The optimum mixture for the nitration mix ranges as follows: It was thought that the contributing acid (sulfuric) aids in the nitration reaction by swelling the fibers so that the nitrate groups could penetrate the fibers more easily and evenly. Nitrate ester formation is favored over sulfate ester formation so that comparatively few sulfate groups are left bound to the cellulose nitrate. The nitration of cellulose nitrate appears, on paper, to be a fairly simple reaction. However, no clear comprehensive mechanism has been found to describe it. Analysis of carefully controlled and nitrated cellulose indicates that the reaction is not as simple as was believed. The percentage of nitrogen in the resultant cellulose nitrate indicates that the reaction is not stoichiometric. The irregular nitration was thought to be a result of inhomogeneities in the original cellulose fibers (Clark 1981, 247). It was believed that the amorphous and crystalline regions of the cellulose fibers reacted differentially to the nitration mix. The comparatively large sulfate molecules were believed to enter the amorphous regions, creating pathways for the smaller nitrate groups. The sulfate groups were supposedly too large to enter the crystalline regions, where the nitrate groups had to fend for themselves. This model would indicate that NO3 and SO4 concentrations or percentages would vary randomly throughout a fiber and might explain nonstoicheometric nitration. A study by D. T. Clark (1981, 247–49) indicates that the physical structure of cellulose may not be responsible for the irregular nitration of the molecule. Clark's work shows that the degree of substitution of nitrogen—a measure of the percent nitrogen—in cellulose nitrate is 2.8 molecules of nitrogen per cellulose residue. This is not stoichiometric, as noted by earlier chemists. Clark summarizes early concepts on cellulose nitrate by indicating that irregular nitration was thought to be due to inhomogeneities in the structure of cellulose or inhomogeneities in the nitrating mix. To find more concrete proof for the reason for the irregular nitration of cellulose, Clark used electron spectroscopy for chemical analysis (ESCA) to look at the chemical compositions of various regions of cellulose nitrate fibers. Clark varied the depth of the particle beam to obtain the chemical composition at different depths in a cellulose nitrate fiber. He found that the degree of substitution of nitrogen for the whole fibers he examined was the same as The inhomogeneities of cellulose nitrate fibers and the presence of sulfate esters and groups in cellulose nitrate fibers create sources of potential deterioration in the final product. Sulfate esters can hydrolyze to sulfuric acid. 2.4 RINSING, DRYING, KNEADINGAfter the cellulose nitrate fibers were nitrated to the desired percent nitrogen, the acids were drained away and the mass of cellulose nitrate was “quenched” or rinsed with water. This step, introduced commercially in 1865, was considered to stabilize the cellulose nitrate by removing the bulk of the free acids from the mass. Different rinsing procedures abounded, but most involved washing the cellulose nitrate in water at elevated temperatures and/or pressures (Worden 1911, 2:595–96). It was believed that much of the instability of cellulose nitrate was due to free sulfuric and nitric acids trapped in the amorphous regions of the cellulose fibers when the mass of cellulose nitrate was rinsed in water. These trapped free acids could provide a source of deterioration in celluloid. After rinsing, the cellulose nitrate was dried, usually with heat or alcohol, until the water content was less than 2% (Worden 1911, 2:599–605). At this point the cellulose nitrate was kneaded in a machine much like a commercial bread kneader (Worden 1911, 2:606–609). During the kneading, a 50% solution of camphor in alcohol was added to the cellulose nitrate (Miles 1955, 207). The camphor-alcohol solution changes the order or macro-structure of the cellulose nitrate fibers. The molecules are forced apart and eventually lose any regular relationship with other molecules. This reaction is much like the denaturation of proteins by hot water. After the camphor-alcohol is mixed with cellulose nitrate fibers the resultant celluloid plastic has no fibrous orientation of molecules and is called a gel, or as Miles (1955, 206) describes it, after gelation there is “no trace of solid structure” in the celluloid. The reaction is not well understood, but it is known that a homogenous gel will form with one molecule of camphor for each glucose unit (Petitpas and Mathieu 1946, 525). The molecules are not chemically “bound” to each other; this allows for the sublimation and migration of camphor out of and through celluloid. Kneading produces a doughy mass, to which any colorants, fillers, or other ingredients needed in the final celluloid are added. These additives all affect the stability of celluloid. Certain fillers such as TiO2 (Worden 1911, 2:624) are known to reduce the effects of photodegradation of plastics, while others seem to increase the rate of deterioration (Sirkis 1982). The additives kneaded into the celluloid in this step introduce sources of potential deterioration. 2.5 SEASONING, CRYSTALLITE FORMATIONWhen the mixing was complete the mass of celluloid was pressed into blocks of various sizes (Miles 1955, 207). The pressure used to prepare the blocks, and pressure applied later during fabrication, forced the cellulose nitrate molecules together. In areas, the intermolecular camphor molecules were compressed and “squeezed out,” leaving cellulose nitrate molecules again in intimate contact with each other. At sites where this occurs crystallites are formed. These local sites of crystal formation form the centers of crystallization in aging celluloid. The blocks were “aged” in drying sheds for varying amounts of time. This “seasoning” step was required because the celluloid went through dimensional 2.6 FABRICATIONThe fabrication of celluloid objects was a complex process. Celluloid was machined like wood (sawed, drilled, carved, and planed) and was formed with heat and pressure and blow molded. The technique of blow molding was developed for use with celluloid. (The first blow molded objects were small celluloid dolls later known as Kewpie Dolls [DuBois 1972, 44–45].) Elaborate procedures were implemented to create detailed and highly accurate imitations that are deceptively difficult to differentiate from the natural materials. For example, the intricate pattern of lines characteristic of ivory and called the lines of Retzius were imitated fairly accurately. Thin sheets of celluloid were prepared in two colors, one opaque and the other more transparent. The opaque ivory-colored sheets were layered between more transparent sheets of the same color. A block was created out of these alternating layers with heat and pressure. This block was then sliced and the thin slices pressed into another block. The second block, when fabricated, created an accurate imitation of ivory (Worden 1911, 2:683–87). Even at a magnification of 12x structures of celluloid ivory may be indistinguishable from those of real ivory. Another similar example is the imitation of horn. Cones of celluloid were cut from blocks of varying opacity and color. These cones were stacked and pressed into a block (Worden 1911, 2:687–89). When the solid block was machined in a certain manner, accurate striations and laminae were visible. At 12x magnification, imitation horn may be indistinguishable from horn. The details of an object's fabrication can cause deterioration problems. Combs were often made by laminating two layers of celluloid together. The layers were often different colors and opacities and hence different compositions of celluloid. The expansion and/or contraction of the different layers may cause the warping or splitting off of a layer. Auxiliary materials added in the fabrication of objects such as metal fittings and decorative elements create complex interactions during decomposition. 2.7 SOURCES OF INHERENT DETERIORATIONIn summary, the synthesis and fabrication of celluloid objects introduces many potential problems for celluloid as it ages. These inherent problems include:
|