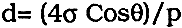A STUDY OF THE DETERIORATION OF EGYPTIAN LIMESTONE SCULPTURES.M. Bradley, & A.P. Middleton
ABSTRACT—The decay of some Egyptian limestone sculptures during storage in a museum environment has been investigated using a variety of analytical techniques. These have included the chemical determination of soluble salts, gravimetric estimation and analysis by X-ray diffraction of the acid insoluble fraction, petrographic examination, observation and analysis in the scanning electron microscope and determination of porosity using mercury porosimetry. The results suggest that the mineralogy of the limestone, in particular the clay content, and the level and nature of soluble salts, particularly of soluble nitrates, are important factors in determining the nature and degree of any decay. The results also indicate that different types of limestone were used at different sites which accounts for the high proportion of badly deteriorated sculpture attributed to sites around Thebes and Abydos. This also suggests a possible technique for determining the provenance of Egyptian limestone objects. 1 INTRODUCTIONTHE BRITISH MUSEUM has an important collection of Egyptian limestone sculptures. Since 1966 this collection has been the subject of a programme of conservation which has included cleaning, consolidation and repair 1–2. A comparison of the present condition of the sculptures with photographs taken when they were added to the collection in the late nineteenth and early twentieth century showed that a small number of sculptures had undergone considerable deterioration in the intervening period (Figures 1 and 2). These sculptures have been stored and displayed in a variety of locations in the museum but, because of a lack of detailed environmental records, it is not possible to relate the degree of deterioration to conditions of storage or display.
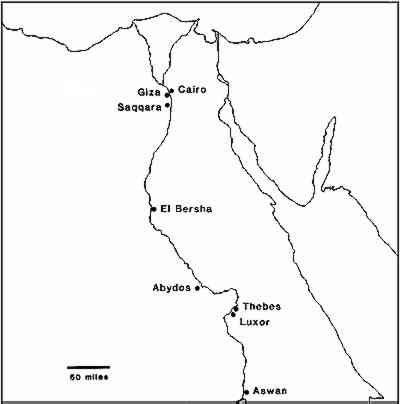
The adverse effect of changes in relative humidity (RH) and subsequent salt movement in contaminated stone have been well documented 3–7, and it is possible that the now badly deteriorated sculptures could have suffered most of this damage from exposure to unusually adverse conditions during or after their removal from the dry conditions of Egypt to the damp maritime climate of England. However it seemed unlikely that this was the only factor involved, since the majority of the sculptures in this collection are still in good condition. It was thought that the chemical and physical properties of the stone itself might render it more or less liable to decay. Factors other than salt content which have been identified as indicators of the durability of the stone include the nature and proportion of clay content 8–10, and pore size distribution, in particular the proportion of the micropores, less then 5 μm diameter 11–13. To investigate the problem, a group of sculptures was selected for detailed examination and analysis; the sculptures included some which had suffered extensive deterioration in the Museum as well as some showing little or no evidence of decay. On examination of the Museum records it was found that the sculptures were attributed to three geographical areas: Thebes and Abydos; El Bersha; and sites around Giza and Saqqara, close to Cairo (Figure 3). The consistency of the analytical
TABLE 1 Details of Sculptures The investigation involved in the analysis of soluble salts, the determination of the amounts of acid insoluble matter, X-ray diffraction analysis (XRD) of the acid insoluble matter, petrographic examination of polished thin sections, and scanning electron microscopy on polished thin sections and fracture surfaces. In addition, porosity determinations were made on a number of samples using mercury porosimetry. 2 EXPERIMENTALTWO SAMPLES WERE TAKEN from each sculpture, one by drilling 50mm into the body of the stone using a 3mm diameter drill bit in a hand drill. The first 1mm of the drilling was discarded to avoid contamination. This sample was used for analysis of soluble salts and for the determination of acid insoluble matter. For the second sample, a small fragment of stone was removed from the underside or back of the sculpture. From this fragment a polished thin section for optical and scanning electron microscopy and also a fracture surface for examination in the SEM were prepared. When required, samples for mercury porosimetry were also obtained from these fragments. 2.1 Soluble salt analysisA portion of the powder obtained by drilling the sculpture was weighed in to a 25ml volumetric flask and distilled water added to the mark. The sample was left to soak for at least 24 hours before the mixture was filtered. Soluble salt analysis was carried out on the aqueous extract using specific ion electrodes with a specific ion meter to measure soluble chloride and nitrate levels. The acidified barium chloride spot test was used to test for the presence of sulphate ions 14. The results of the chloride and nitrate analyses were corrected for variations in solution temperature and were calculated as weight/weight percent soluble salt to stone sample. 2.2 Acid insoluble matter determinationThe proportion of acid insoluble matter was determined by weighing a portion of the drilling into a beaker and adding 1M hydrochloric acid. The mixture was left to react for one hour at ambient temperature and then filtered under vacuum through a pre-weighed Gelman DM450 membrane filter. The filter was allowed to dry to 2.3 X-ray diffraction analysisAcid insoluble residues and stone powder were analyzed by X-ray diffractometry (XRD) using nickel-filtered copper radiation with a Philips PW1050 goniometer and PW1710 controller. Samples of air dried, acid insoluble matters were analyzed directly on the membrane filters prepared as above (2.2.). A selection of these samples was also analyzed after exposure to ethylene glycol vapor (48 hours, 55�C) in order to determine whether the clays had expanded (see reference 19). Portions of some of the undigested stone powder samples were ground in an agate pestle and mortar, dispersed in distilled water in an ultrasonic tank and similarly deposited on membrane filters for analysis. 2.4 Petrographic examinationPolished thin sections were prepared from the fragments of stone taken from the sculptures and were examined using a Nikon Optiphot petrological microscope at 20–400x magnification. Uncovered polished thin sections were used rather than the normal covered sections to allow subsequent examination and analysis of the sections in the SEM. 2.5 Scanning electron microscopy and X-ray micro-analysisPolished thin sections and fracture surfaces from the fragments sampled were examined in a JEOL JSM-840 scanning electron microscope (SEM) fitted with a Link Systems model 500, series II energy dispersive X-ray analyzer (EDXA). The samples were coated with carbon to make them conducting and to facilitate elemental analysis of features identified morphologically. 2.6 Mercury PorosimetryMercury porosimetry measurements were carried out by the Hydrology Research Institute. Small samples of stone, approximately 10mm � 5mm � 5 mm were taken from a total of seven sculptures - five from Thebes/Abydos (three “deteriorated,” two “undeteriorated”) and one each from Saqqara and Giza. The samples were dried at 60� C to a constant weight before being placed in the sample holder. The pressure in the sample holder was reduced to 1 PSI and mercury gradually introduced into the specimen with increasing pressure up to 35000 PSI. The intrusion of mercury into
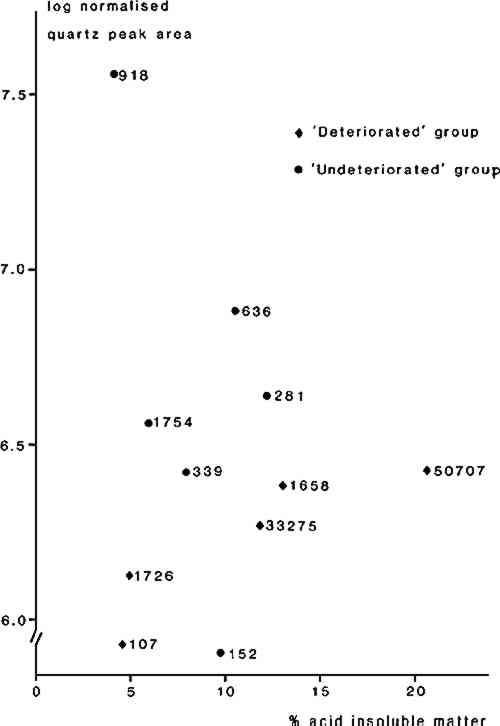
3 RESULTS3.1 Soluble salt analysisTESTS OF THE AQUEOUS extracts for the presence of sulphate ion were negative for all samples. Both chloride and nitrate were present in the “deteriorated” samples from Thebes/Abydos, but in several of the “undeteriorated” sculptures only chloride ions were detected, the level of nitrate ions being below the limit of detection (ca. 1ppm). Statistical analysis using a non-parametric test 17 showed that the nitrate levels tend to be significantly higher in the “deteriorated” group of sculptures but there was no statistical difference in chloride levels between the two groups. The soluble salt levels determined for sculptures from the Cairo area are similar to those for the “undeteriorated” group from Thebes/Abydos. The sculptures from El Bersha were found to have very low levels of both chloride and nitrate. The results are given in Table 2. TABLE 2 Summary of Analytical Results 3.2 Acid insoluble matter determinationThe results of the acid insoluble matter determination are also given in Table 2. The samples from El Bersha have very low acid insoluble fractions whilst those from Cairo and Thebes/Abydos generally have high acid insoluble fractions, indicating differences in the mineralogy of the stones used at the various locations. Although the mean acid insoluble fraction level for the “undeteriorated” group from Thebes/Abydos is lower than for the “deteriorated” group of sculptures there is considerable overlap in the results and the two groups could not be distinguished by statistical tests. 3.3 Identification of acid insoluble fraction (XRD, EDXA)The X-ray diffraction traces obtained on the acid insoluble fractions showed differences between the objects; three broad categories of residue were recognized. These appeared to correlate with the geographical provenance of the sculptures. The results are summarized in Table 2. There was considerable similarity between the acid insoluble fractions obtained from the “undeteriorated” and “deteriorated” sculptures from Thebes/Abydos, and the XRD results indicated that they generally consist of a mixture of non-expanding clay and quartz. Analysis of the clay in the SEM, using EDXA, showed that in most samples it is a magnesium silicate, very low in aluminum, consistent with its identification as either a member of the sepiolite-palygorskite group or as a low aluminum smectite such as hectoraite or stevensite.19,20 The XRD data, particularly the apparently non-expanding nature of the clay, suggest that it is probably an aluminum-poor member of the sepiolote/palygorskite group, since smectite minerals would have been expected to expand on exposure to ethylene glycol vapor. In a few samples, those from EA 33275 and EA 636, the XRD traces indicated that the clay was not of this type, and analysis using EDXA showed that these clays contained aluminum, potassium and iron in addition to magnesium and silicon. Examination of the XRD traces indicated that, in general, the quartz content of the acid insoluble fraction in the “undeteriorated” sculptures from Thebes/Anydos was higher than in residues from the “deteriorates” sculptures. This was tested for the samples from Thebes/Abydos (exception EA 805 for which insufficient sample was available) by determining the quarts peak areas (101 peak) and plotting values normalized for sample weight against the percentage of acid insoluble fraction in the limestone (Figure 6). Statistical analysis of these data, using a non-parametric test, confirmed the significance (at the 95% level) of this difference between the two groups of sculptures.
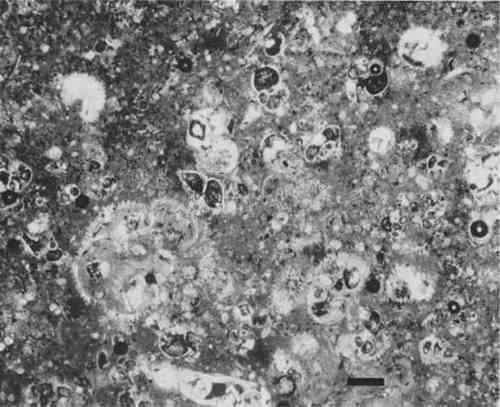
X-ray diffraction analysis was also carried out on stone powder from some of the sculptures from Thebes/Abydos. The traces obtained showed the presence of dolomite in the samples, and normalized peak area measurements (104) peak were obtained for dolomite, as described above for quartz. Although initial inspection of the values suggested that more dolomite was present in the samples from the “deteriorated” group of sculptures, subsequent statistical analysis did not confirm this. X-ray diffraction patterns from the acid insoluble fraction of the sculptures from Saqqara and Giza indicate that they consist mainly of opal-CT, a metastable form of silica,18 sometimes with minor amounts of quartz and/or clay. The very low acid insoluble fractions of the limestone from El Bersha were identified as being mainly quartz. 3.4 Petrographic examinationPetrographic Examination of the polished thin sections indicated that most of the samples are fine grained micritic limestones containing a few fossil fragments (fossilifereous micrites, 21), although some are highly fossiliferous biomicrites (Figure 7). In general it was not possible to observe the non-carbonate fraction in any of the sections in detail, although some EA 33275 from Thebes exhibits form birefringence due to the strongly developed preferred orientation of the clay platelets. The limestones from Thebes and Abydos generally show evidence for partial dolmitisation in the form of small rhombohedra of dolomite (Figure 8). No significant differences were observed between the “deteriorated” and “undeteriorated” groups of sculptures from Thebes/Abydos.
3.5 Scanning electron microscopyThe polished thin sections and fracture surfaces from the sculptures were examined in the SEM. Although in the fracture surfaces detail such as the presence of sodium chloride crystals, grain morphology and distribution and morphology of the non-carbonate fraction were visible, most useful information was derived from the polished thin sections. The observations revealed significant differences between the sculptures, dividing them into three groups which correlate with the geographical provenance. The sculptures from Thebes/Abydos are characterized by the presence of subhedral to euhedral rhombohedra of dolomite in a fine grained micritic matrix of low magnesium calcite (Figure 9). In these samples porosity and the non-carbonate, clay fraction are apparently fairly uniformly distributed throughout the stone. In fracture surfaces the clay could be seen as coatings of flakey or fibrous material on the surface of the calcite grains. The sculptures from Giza and Saqqara have a radically different texture to those from Thebes/Abydos, with islands of relatively solid calcite separated by areas of higher porosity containing lepispheres of silica (opal-CT) (Figure 10). A similar texture was shown by the sculptures from El Bersha (Figure 11), but in these the acid insoluble fraction, present at very low levels, was not readily apparent in the SEM.
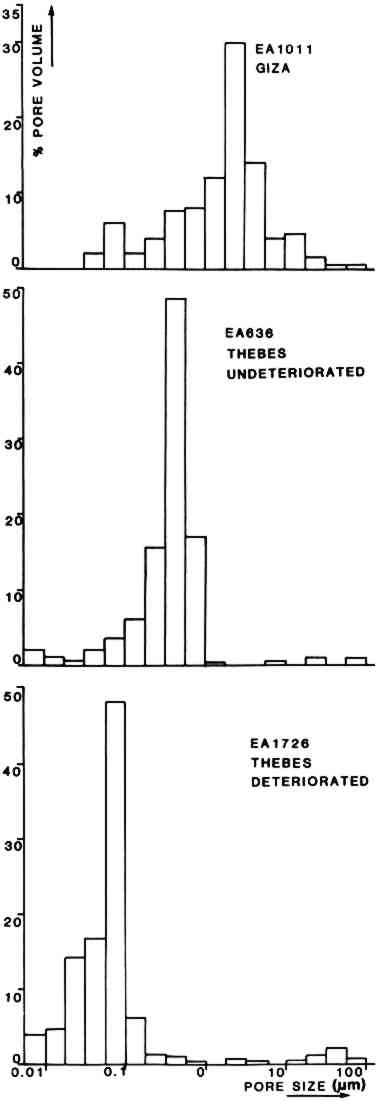
3.6 Mercury PorosimetryHistograms showing pore size distributions of EA 1726 (Thebes, “deteriorated” group), EA 636 (Thebes, “undeteriorated” group) and EA 1011 (Giza) are presented in Figure 12. The data obtained from the determinations are summarized in Table 3. These results suggest that the “undeteriorated” sculptures from Thebes/Abydos have higher total porosity and also higher median pore throat sizes than the “deteriorated” group. Sculptures from the Cairo area have somewhat higher values still of porosity and median throat size. All of the sculptures tested were found to have a high proportion of micropores (i.e. those less than 5 μm in diameter).
TABLE 3 Results of Mecury Porosimetry Determination 4 DISCUSSIONTHE RESULTS OF THIS STUDY indicate that the chemical and physical properties of the various limestones used for the sculptures have exerted a strong influence upon the degree and mode of their subsequent decay in museum storage. A major factor leading to decay appears to be the presence of a relatively high proportion of acid insoluble materials, which consists in large part of clay (see Table 2). The deleterious effects of high levels of acid insoluble material have been recognized for some time, 1–2 and the effects of clay in particular on stone strength and durability have also been noted. 8–10,13 The results of the present investigation indicate that the mineralogical character of the acid insoluble fraction is of crucial importance in determining the durability of the stone. Thus, although the mean acid insoluble content of the relatively well preserved sculptures from Giza and Saqqara is high (x� = 5.8%), it is present as silica (opal-CT), which is likely to be a more stable component than those found in the sculptures from Thebes/Abydos. Another factor which appears to be significant is the presence of high levels of soluble salts, especially when these include a high The importance of high levels of nitrate in rendering the stone susceptible to decay is indicated in the present results for the “deteriorated” and “undeteriorated” sculptures from Thebes/Abydos. All of the objects studied contain high levels of soluble chlorides, above the 0.1% threshold of stability established by Oddy et al., 1 and the “deteriorated” and “undeteriorated” groups are indistinguishable statistically. However, as noted above (section 3.1), statistical analysis of the results for soluble nitrate shows that, on average, the “deteriorated” group have significantly higher levels than the “undeteriorated” group. A third factor which appears to be significant is the petrographic character of the limestone. Those from Thebes/Abydos have textures in which the calcite is highly fragmented, occurring as dispersed grains and aggregates separated by fine grained clay and open pores, providing easy access for moisture, as well as greater surface area for chemical reaction. These rocks also contain subhedral-euhedral crystals of dolomite but the significance of these to the decay process is not known. In contrast, the better preserved sculptures from Cairo and from El Bersha exhibit less fragmented textures, with solid islands of calcite separated by regions of higher porosity (see section 3.5). It is thus possible to identify for each of the three groups of sculptures particular features which have influenced their mode and extent of the decay. 4.1 The Thebes/Abydos groupThis group of sculptures includes those which exhibit the most severe decay. This seems to be because of the combination of several adverse features, inherent in the stone, viz. high acid insoluble fractions which are predominantly of clay; high soluble salt contents which are high in nitrate; and a highly fragmented petrographic texture. Reasons for the observed differences in condition between the “deteriorated” and “undeteriorated” sculptures from this area are less easy to pinpoint. One factor, concerning which we have no information, is the past conditions of storage of the objects; it may be, at least in some cases, that the present condition of the sculptures reflects their previous storage history. However, the analytical results do suggest that several other factors may also be significant, and it appears likely that a combination of high soluble nitrate content, high clay content, and high microporosity, perhaps exacerbated by adverse environmental conditions, has resulted in the advanced deterioration observed in some samples. Observation of the chemical and physical properties of the stone may thus be useful in identifying those sculptures from Thebes/Abydos which are most at risk. The statistical technique of discriminant analysis 24 would be well suited to this 4.2 The Giza/Saqqara groupThe decay pattern observed in the sculptures from around Cairo (Giza and Saqqara) is one of pitting, powdering and some flaking, the body of the stone being firm and undeteriorated. The relative soundness of these sculptures is thought to be attributable to several factors, including the presence of only low levels of nitrate, despite relatively high levels of chloride (see Table 2); and a relatively solid, unfragmented petrographic texture. Surface powdering of these pieces has perhaps been caused by the movement of soluble salts close to the surface of the sculptures, the minor pitting observed may result from localized chemical attack in the regions of higher porosity between the more solid islands of calcite. 4.3 The El Bersha groupThe sculptures from El Bersha, which show only minor pitting are very pure calcite limestones with low acid insoluble fractions and low soluble salts and they therefore fit well with previously established criteria for limestone durability. 1,2 As for the Giza/Saqqara sculptures, the minor pitting which is observed probably results from localized chemical attack in the regions of higher porosity between the more solid islands of calcite. 5 CONCLUSIONSTHIS INVESTIGATION OF EGYPTIAN sculpture has demonstrated that a number of factors, especially in combination, may be important in determining the durability of limestone objects.
As noted above, the sculptures from Thebes/Abydos, because of the particular properties of the limestones used are most at risk amongst those included in this study. The deterioration of sculptures of this type, which are of low durability, can be minimized by controlling environmental conditions in storage and display areas. This technique has been used successfully on another collection of sculptures in the Museum which are now stored under conditions of constant relative humidity and temperature to avoid wet-dry cycles affecting clays and to avoid An area of study which this investigation has incidentally touched upon is the determination of provenance for Egyptian limestone sculpture. An analytical approach, using neutron activation analysis, was described by Meyers and van Zelst25 and their results permitted a distinction between limestone sculptures from around Theses and those from further north. The present study has shown that several petrographically distinct types of limestone were used for Egyptian sculpture and that these can be characterized by several analytical techniques, especially X-ray diffraction and scanning electron microscopy. Possible implications for the determination of the provenance of Egyptian limestone, using these techniques, are being actively explored. ACKNOWLEDGEMENTSWE ARE GRATEFUL to several of our colleagues for their constructive comments and discussion throughout this project, especially Dr. I.C. Freestone, Dr. M.N. Leese, Mr. W.A. Oddy and Dr. M.S. Tite. We are also grateful to Dr. M. Price of the Hydrogeology Research Institute, British Geological Survey, Wallingford, England for organizing the pore size distribution measurements on our behalf, and Dr. H.F. Shaw of Imperial College, University of London for assistance with the interpretation of the XRD data. We would like to thank members of the Department of Conservation, Stone Section for their help in sampling the objects and Mr. T.G.H. James, Keepter of the Department of Egyptian Antiquities and his staff for their support and cooperation throughout this project. NOTES. 1Oddy, W.A., Hughes, M.J. and Baker, S., “The Washing of Limestone Sculptures from Egypt and the Middle East”, Lithoclastia, No. 2(1976) 3–10. . 2Barton, N.G., and Blackshaw, S.M., “Statistical Evaluation of the Analyses Carried Out on Egyptian Limestone”, Lithoclastia, No 2 (1976) 11–16. . 3Winkler, E.M., Stone: Properties and Durability In Man's Environment, 2nd Edition, Springer Verlag, New York (1975)119–125. . 4Lucas, A., Disintegration and Preservation of Building Stones in Egypt, Ministry of Fianance, Egypt, Cairo (1915). . 5Winkler, E.M., Stone:Properties and Durability in Man's Environment, 2nd Edition, Springer Verlag, New York (1975) 102–125. . 6Arnold, A., and Zehnder, K., “Crystallisation and Habits of Salt Efflorenscences on Walls II: Conditions of Crystallization,” Proceedings 5th International Congress on Deterioration and Conservation of Stone, Lausanne (1985) 269–278. . 7Binda, L., Baronio, G., and Charola, A.E., “Deterioration of Porous Materials Due to Salt Crystallization Under Different Thermohygrometric Conditions,” Proceedings 5th International Congress on Deterioration and Conservation of Stone, Lausanne (1985) 279–288. . 8Bromble, P., and Bocquier, G., “Donnees Petrologiques Concernant L'alteration Des Gres Des Temples De Karnak (Egypte),” Proceedings 5th International Congress on Deterioration and Conservation of Stone, Lausanne (1985) 361–370. . 9Ordaz, J., and Esbert, R.M., “Porosity and Capillarity in Some Sandstone and Dolomite Monumental Stones,” Proceedings 5th International Congress on Deterioration and Conservation of Stone, Lausanne (1985) 93–102. . 10Abd el Hady, M.M., and Krzywoblocka-Laurov, R., “The Durability of Limestone Employed in Roman Theatre and Qait Bay's Citadel in the Marine Environment in Alexandria Egypt,” Proceedings 5th International Congress on Deterioration and Conservation of Stone, Lausanne (1985) 307–312. . 11Schaffer, R.J., The Weathering of Natural Building Stones, Building Research Report No. 18, Building Research Establishment, Garston (1972) 33–36. . 12Honeyborne, D.B., and Harris, P.B., “The Structure of Porous Building Stone and its Relation to Weathering,” Volume 10, Proceedings of the Colston Research Society, Butterworths (1958) 343–365. . 13Leary, E., “The Builiding Limestones of the British Isles,” Buidling Research Establishment Report, HMSO (1983) 6–8. . 14Bassett, J., Denney, R.C., Jeffery, G.H., and Mendham, J., Vogel's Textbook of Quantitative Inorganic Analysis, 4th Edition, Longman, London (1978)504–507. . 15Experimental Methods, Test No. 1.5, Proceedings of International Symposium on the Deterioration and Protection of Stone Monuments, UNESCO-RILEM, Vol.5, Paris June 5–9, 1978. . 16Wardlaw, N.C., and McKellar, M., “Mercury Porosimetry and the Interpretation of Pore Geometry in Sedimentary Rocks and Artificial Models”, Powder Technology, 29 (1981) 127–143. . 17Siegel, S., Non Parametric Statistics of the Bahavioral Sciences, McGraw-Kogakusha, Tokyo, (1956) 312 pp. . 18Calvert, S.E. “Marine Mineralogy - Mineralogy of Silica Phases in Deep Sea Cherts and Porcelanites,” Philosophical Transactions of the Royal Society of London, A286 (1977)239–252. . 19Brindley, G.W., and Brown, G., (editors) Crystal Structures of Clay Minerals and their X-ray Identification, Mineralogical Society, London, (1980). . 20Hassouba, H., and Shaw, H.F., “The Occurence of Palygorskite in Quaternary Sediments of the Costal Plain of North West Egypt,” Clay Minerals, 15 (1980) 77–83. . 21Folk, R.L., “Spectral Subdivision of Limestone Types,” in Classification of Carbonate Rocks, (ed. W.E. Than), Memoir of American Association of Petroleum Geologists, 1 (1962) 62–84. . 22Arnold, A., Kung, A., and Zehender, K., “Deterioration and Preservation of Carolingian and Medieval Mural Paintings in the Mustair Convent (Switzerland) Part 1 Decay Mechanisms and Preservation,” in Case Studies in the Conservation of Stone and Wall Paintings, (ed. N.S. Brommelle, and Perry Smith), IIC, Bologna (1986) 190–194. . 23Arnold, A., and Kung, A., “Crystallization and Habits of Salt Effloresences on Walls 1: Methods of Investigation and Habits,” Proceedings of Fifth International Congress on Deterioration of Stone, Lausanne (1985) 255–267. . 24Cooley, W.W., and Lohens, P.R., Multivariate Data Analysis, New York, (1971). . 25Mayers, P., and Van Zelst, “Neutron Activation of Limestone Objects: A Pilot Study,” Radiochimica Acta, 24 (1977) 197–204. . 26Unpublished work carried out at the British Museum on a group of partially metamorphosed limestone sculptures by S.M. Bradley, I. Freestone, W.A. Oddy and A.E.A. Werner.
 Section Index Section Index |