ON THE REVERSIBILITY OF TREATMENTS WITH ACRYLIC/SILICONE RESIN MIXTURESA.E. Charola, A. Tucci, & R.J. Koestler
ABSTRACT—The reversibility of a consolidation treatment on limestone, using an acrylic/silicone mixture, is tested on aged and unaged specimens. Extracted residues are tested by infrared analysis and scanning electron microscope examination. Irreversibility of the treatment is found to be confined to the exposed surface of the stone. 1 INTRODUCTION“IN CONSERVATION WORK, there is a dictum that nothing should be done that cannot, if necessary, be undone easily in the future.” This statement by Plenderleith1 sums up the importance of the reversibility of any treatment applied. That reversibility is one of the most desirable features cannot be denied. Yet the broadness of the term “treatment,” which could include cleaning, gluing, consolidating, etc., makes this issue a very debatable one. For instance, reversibility is rarely mentioned as a consideration in the cleaning of stone, whereas the concept has acted as a severe handicap to the testing of consolidants. Furthermore, it has also allowed for different interpretations of the meaning of “reversibility.” For example, Sanpaolesi interpreted the reversibility of the treatment as a philosophy to be applied to the overall conservation/restoration of a monument; but not to be taken literally when it came down to the actual consolidation of the deteriorating material.2 In the search for a “reversible” consolidation treatment, the more important principle, that any applied treatment should not interfere with future necessary treatments, as mentioned by the 1972 Italian Carta del Restauro3 and given as one of the three conservation principles by Brandi,4 has been generally forgotten. It must also be remembered that “total reversibility” is a theoretical concept which is not found in nature. We propose that total reversibility should be limited to the possibility of complete removal during the application of the treatment, to allow for esthetic adjustments.5 The whole rationale for consolidation is based on the fact that the material is deteriorating so badly that unless it is “treated” it will not survive. The “treatment” will not safeguard the material from further deterioration but will hopefully slow down the deterioration process and give the object a longer life so that in the future a new treatment can be applied if necessary.6 Consolidating and water-repelling treatments based on mixtures of acrylic and silicone resins have been fairly widely used in recent years. In particular, the mixture of Acryloid B72 and DriFilm 104 has found extensive application in Italy.7–9 Because of its wide use it has been subjected to careful laboratory studies.10–12 During these studies it was found that acid rain modified the microstructure of the silicone resin on This paper studies the difference in extraction facility of weathered specimens versus unweathered ones, i.e., the reversibility of the treatment after ageing has occurred. 2 EXPERIMENTAL2.1 Sample PreparationTwo limestones were used for this study, an oolitic one, Indiana limestone, and a fossiliferous one, from Vicenza, Italy. Cubic samples (5 � 5 � 5 cm) were treated with a mixture of acrylic and silicone resins. The samples were treated by capillary rise with the following mixture:
2.2 Accelerated WeatheringSome of the samples were weathered by repeated cycles of sulphuric acid fog (4 hours) followed by drying in a climatic chamber (20 hours). The samples were subjected to 21 cycles in total. Acid fog was obtained by means of a 0.02 M H2SO4 solution. About 0.01 ml/cm2 of acid solution collected per hour on the horizontal face of the sample (the face that had been in contact with the mixture during the treatment, referred to from here on as the top face). The climatic chamber was equipped with a 125 W UV lamp, of high pressure mercury vapor, with highest emission at 280–380 nm, which was left on during the length of the exposure in the chamber at 50�C and 70% RH. More details on the accelerating weathering have been reported previously.11,12 2.3 Extraction of the Applied ResinThe applied resin was extracted from weathered and unweathered specimens by two methods:
2.4 Infrared Analysis of Extracted ResiduesIR spectra were obtained by forming a film on a NaCl plate from the solvent solution obtained from either the poulticing or the total immersion of the sample. 2.5 SEM examinationSamples of specimens of the outer surface of the untreated limestones, treated, and treated and weathered, were examined under SEM. The last two types of samples were also examined after poulticing or immersion extraction had been performed. The specimens were mounted on appropriate stubs and sputter-coated with 10nm of gold. 3 RESULTS AND DISCUSSIONSEM EXAMINATION OF specimens of Indiana and Vicenza limestone showed that there were no differences between the two with respect to the results obtained. Also, the IR spectra of the residues obtained from the poultices and/or immersion of the samples were similar regardless of the limestone from which they had been extracted. The illustration of the data for the discussion will therefore be carried out from specimens of Vicenza limestone. Figure 1 shows the appearance of a specimen of Vicenza limestone after treatment with the resin mixture. The treatment is uniform and covers all of the limestone grains. For comparison, the appearance of an untreated limestone is similar to that shown in Figure 5. The appearance of the treated sample does not change, even after one year, in specimens that are kept in the laboratory.
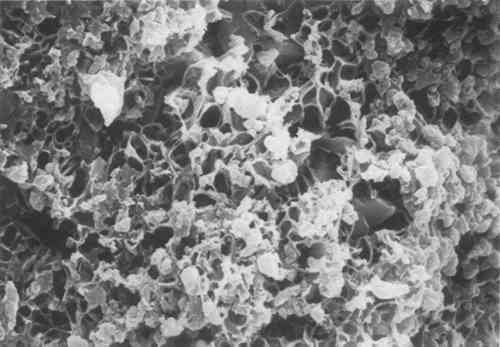
After ageing the specimens in a weathering chamber, the microscopic appearance of the mixture changes significantly, as can be seen in Figure 2. The network of resin formed is more evident because of the acid attack which dissolved some surface grains.
If this sample is then poulticed, the network that remains is much more
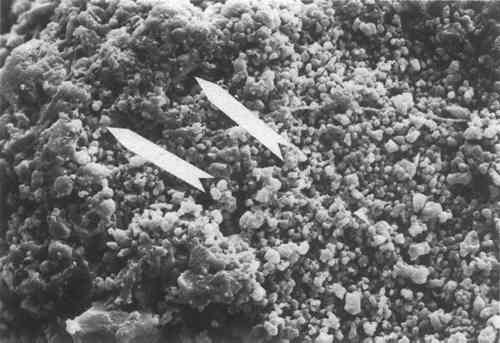
The appearance of an unweathered specimen after poulticing is shown in Figure 4. Some residues of the resin are still visible, but most have been removed. This is the appearance on the poulticed face. On side faces, the appearance is intermediate between that of Figures 1 and 4. Figure 5 shows another unweathered specimen after total immersion in the solvent mixture. The appearance is equivalent to one of an untreated specimen, indicating that the resin has been removed entirely.
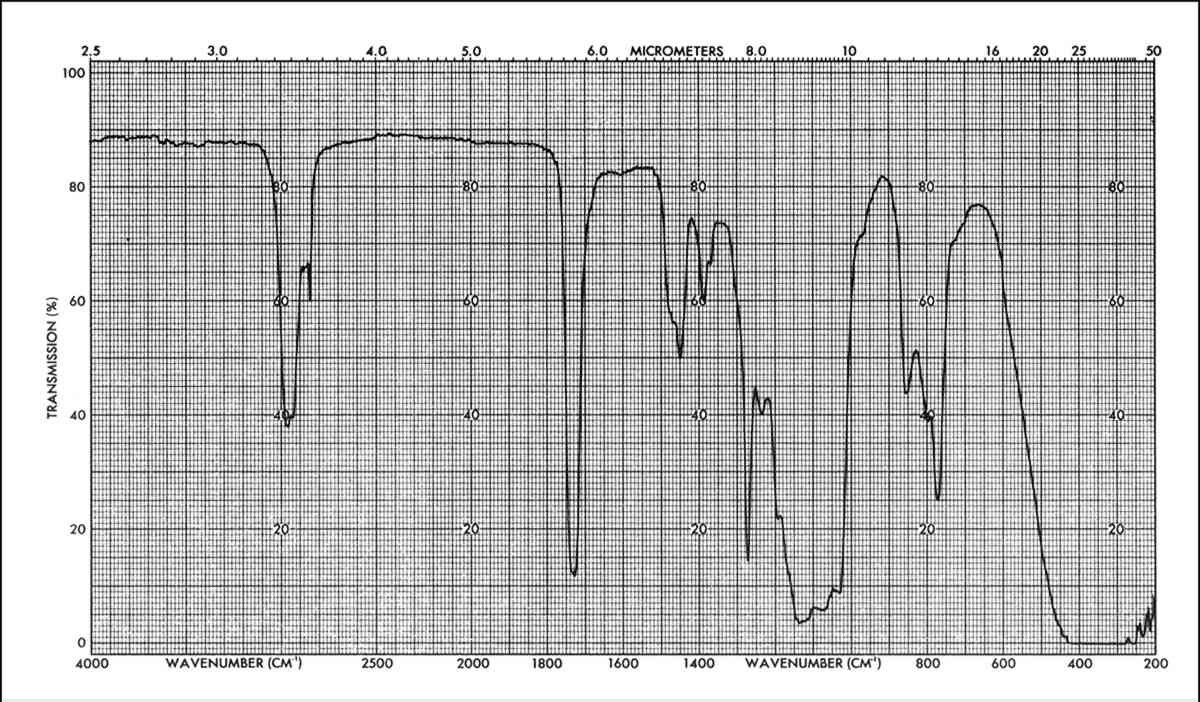
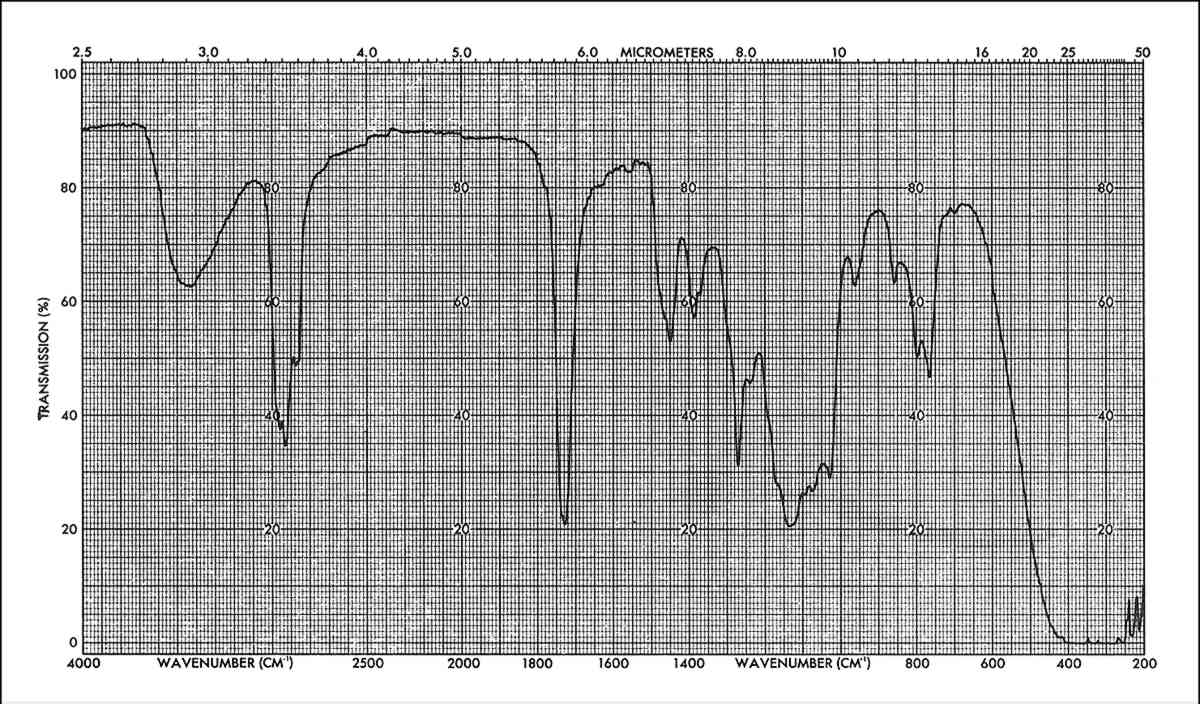
The IR data show that the spectra obtained after total immersion or poulticing of the unweathered specimens are fairly similar to that of the original spectrum of the applied mixture, as shown in Figures 6 and 7. The main difference can be seen in the appearance of the 960 cm−1 peak (corresponding to C-H out of plane bending and/or CH2 rocking), in the relative change of intensities of the C-H stretching bands (around 2900 cm−1) and also with respect to the carbonyl peak (1725 cm−1) and in the relative change of intensities of the three peaks around 800 cm−1. These changes suggest that not all material has been extracted (a partition of different molecular weight polymers can occur in the stone) and/or that some interaction between the components of the mixture has occurred.
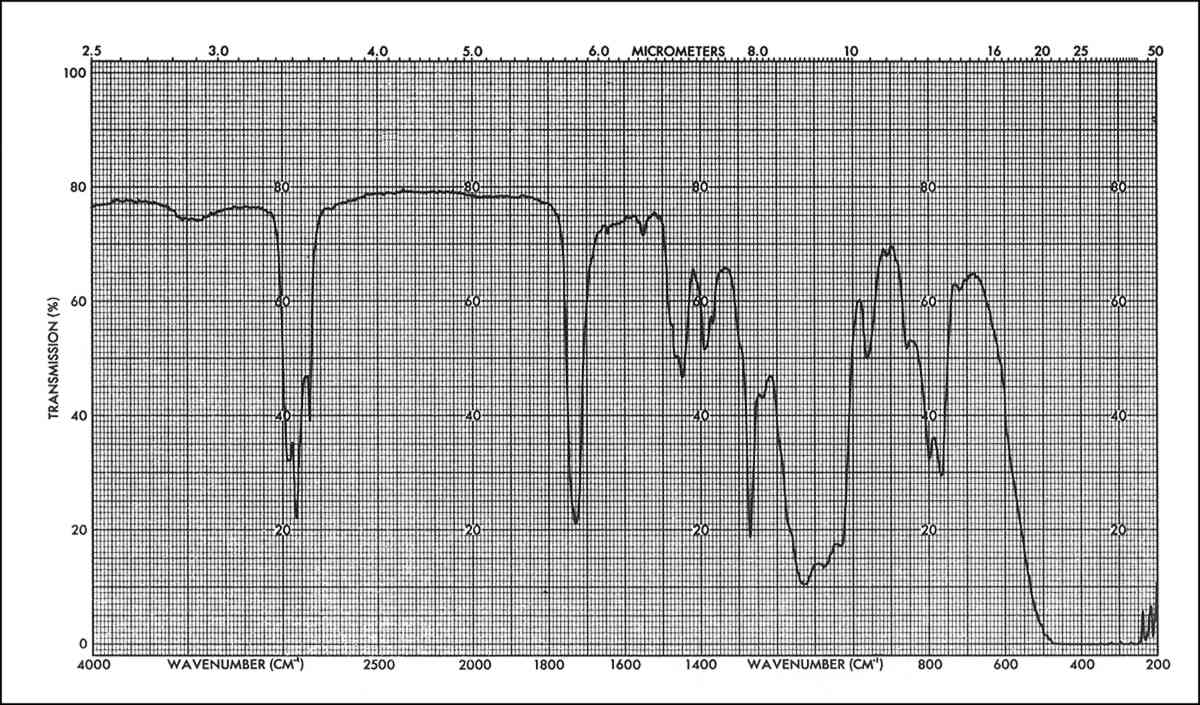
Spectra of the solvent extracts from immersion of weathered specimens is similar to the immersion spectra of unweathered specimens. On the other hand, the resin extracted by poulticing a weathered specimen, Figure 8 is remarkably changed. The main difference is in the relative intensities of the peaks corresponding to the B72 and the DF104; for example, the distortion of the Si-O-Si band at 1050–1150 cm−1 by the influence of the B72 doublet at 1150 cm−1 (corresponding to the C-O-C stretching of the -COOC- group and the C-Ostretching of the -O-CHz-). This indicates that in this case the proportion of B72 to DF104 has changed with respect to the original composition. By calibration with standard mixtures of these two components it was found that the extracted mixture had a composition of about 9:3.5::B72:DF104 while the original mixture applied had a composition of 9:7::B72:DF104. That is, the amount of DF104 in the extracted mixture, after weathering, was half that of the original application.
This change in composition can be explained by the continued polymerization of the silicone resin DF104 through the catalyzing action of the acid rain.11,12 This in-situ polymerization renders this resin more insoluble in solvents. That the solution extracted by immersion from a weathered specimen should be similar to that obtained from an unweathered specimen can be explained by the fact that the in-situ polymerization occurs only on the very surface of the stone.12 When the resin mixture is extracted from the whole specimen, the bulk of this mixture will have the original composition. 4 CONCLUSIONSIT WAS FOUND THAT the consolidating and water-repelling treatment based on mixtures of B72-DF104 is not “totally” reversible once the treatment has aged. Specifically, the in-situ polymerization of the silicone resin makes this part of the treatment irreversible. This irreversibility is limited to the exposed surface of the stone only. This feature should not be a condemning factor, as the surface will be the most weathered part, and should the treatment be totally reversible after the stone has weathered, the surface would be lost because of lack of cohesion. This treatment, based on a mixture of acrylic and silicone resins was found to be totally reversible for the bulk of the treatment, while only a thin surface layer turns partially irreversible. This remaining surface layer, because of its chemical nature and its microstructure, should not interfere with future treatments. MATERIALSAcryloid B72:an acrylic resin copolymer of methyl acrylate and ethyl methacrylate, Rohm & Haas, Philadelphia, PA 19105, USA DriFilm 104:a partially pre-polymerized alkoxysilane, General Electric, Waterford, NY 12188, USA ACKNOWLEDGEMENTSTHE AUTHORS WISH TO THANK Dr. L. Lazzarini and Dr. N. Indictor for helpful comments and suggestions. Also to be thanked is C. Blair for the photographic reproductions of the SEM micrographs. BIBLIOGRAPHYPlenderleith, H.J. “Preservation and Conservation: Introductory Statement” Preservation and Conservation: Principles and Practices (Proceedings of the North American International Regional Conference 1972, xvii–xxi.) Washington, D.C.The Preservation Press, 1976. Gurrieri, F. “Piero Sanpaolesi, II Restauro come Scienza” Piero Sanpaolesi, II Restuaro, dai principi alle tecnicheVI ICOMOS General Assembly, Florence1981, 7–12. Carta delRestauro [1972] Ideologie e prassi del restauro con Antologia di testiG.La Monica, Libreria Nuova Presenza, Palermo1975, 213–230. Brandi, C., Teoria del Restauro Piccola Biblioteca Einaudi, Torino1977, 13–20. Cavaletti, R., Lazzarini, L., Marchesini, L. and Marinelli, G. “A New Type of Epoxy Resin for the Structural Consolidation of Badly Decayed Stones” Proceedings of the 5th International Congress on Deterioration and Conservation of Stone, Lausanne1985, 769–778. Torraca, G. “Brick, Adobe, Stone and Architectural Ceramics: Deterioration Processes and Conservation Practices” Preservation and Conservation: Principles and PracticesProceedings of the North American International Regional Conference, 1972, The Preservation Press, 143–165, 1976. Washington, D.C. Rossi-Manaresi, R., Alessandrini, G., Fuzzi, S. and Pruzzi, R. “Assessment of the Effectiveness of some Preservatives for Marble and Limestone” Proceedings of the 3rd. International Congress on Deterioration and Preservation of StoneVenice1979, 357–376. Rossi-Manaresi, R. “Effectiveness of Conservation Treatments for the Sandstone Monuments in Bologna” The Conservation of Stone II, Proceedings of the International SymposiumBologna1981, 665–688. Rossi-Manaresi, R. “Scientific and Technical Research” Jacopo della Quercia e la Facciata de San PetronioCentro per la Conservazione delle Sculture all'ApertoBologna1981, 225–277. Charola, A.E., Rossi-Manaresi, R., Koestler, R. J., Wheeler, G.E. and Tucci, A. “SEM Examination of Limestones Treated with Silane or Prepolymerized Silicone Resin in Solution” Preprints of the IIC Congress Adhesives and ConsolidantsParis1984, 182–184. Rossi-Manaresi, R., Charola, A. E., Tucci, A., Koestler, R. J., and Wheeler, G. E. “Study of Accelerated Weathering of Limestones treated with an Acrylic-Silicone Mixture” Proceedings of the 7th Triennial Meeting of ICOM, Copenhagen1984101–4. Tucci, A., Koestler, R.J., Charola, A.E. and Rossi-Manaresi, R. “The Influence of Acid Rain and UV Radiation on the Ageing of Acrylic and Silicone Resins” Proceedings of the 5th International Congress on Deterioration and Conservation of Stone, Lausanne1985, 891–898.
 Section Index Section Index |