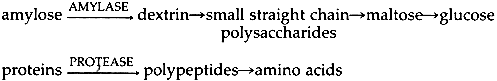SOME OBSERVATIONS ON THE USE OF ENZYMES IN PAPER CONSERVATIONPia C. DeSantis
2 GENERAL TERMINOLOGYENZYMES ARE PROTEINS found in organisms and their purpose is to help those organisms use efficiently the materials they consume. As catalysts, enzymes increase the speed of reactions without being consumed themselves. The enzymes of interest to conservators catalyze degradation reactions, mostly hydrolytic reactions, and usually belong to the class known as hydrolases. The preparations which conservators have purchased in the past have been extracted from the cells of bacteria, molds and cattle pancreas. Like all proteins, enzymes are made up of amino acids linked by peptide bonds between the amino group of one acid and the carboxyl group of the next.1 The chain comprising each enzyme has a unique sequence of amino acids as well as a unique configuration, and their uniqueness makes it possible for hydrolytic enzymes to catalyze the breakdown of only certain molecules.2 These molecules, called substrates,3 fit into the pockets created by the enzyme's twisted chain.4 The ability to enter an enzyme pocket ensures the close interaction of the reacting substrates, thereby lowering the initial energy needed for their reaction. Thus, the degradation of the substrates takes place in a far shorter time than would be necessary without the benefit of the enzyme.5 When in the organism, the enzyme depends on its environment to function properly.6 Temperature and pH shape this environment, and consequently enzyme reactivity and efficiency are temperature and pH dependent.7 Product literature will often refer to temperature and pH optima which supposedly provide the user with conditions for eliciting the enzyme's peak efficiency. However, these terms are not absolute but relative, and their applicability to our purposes will be discussed below. All enzyme molecules have amino acids as part of their structure, but some may also include sugars, vitamins and a number of other compounds which may be necessary for the enzyme's stability or catalytic efficiency.8 Enzyme molecules which include metals are called metalloenzymes and form an important subgroup to be discussed below. At present, two types of enzymes have been found useful in paper conservation: first, the amylases, or starch digesting enzymes, which quicken the degradation of old starch paste, and second, the proteases, or protein digesting enzymes, which speed the degradation of old hide glue. The reaction pathways have been summarized by Grattan, St. Hilaire, Burgess and McCawley:9
|