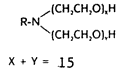Removal of Varnish From Paper Artifacts
by Tatyana PetukhovaThe collections of university libraries and small museums often include drawings or hand-colored maps, varnished originally to protect them but now in a sadly deteriorated state. the treatment of such artifacts presents a challenge for a conservator, particularly when varnish removal must be undertaken to stabilize the artifact. in some cases where removing the varnish coating is the preferable choice, the conservator may not proceed with the treatment because of the risk of the partial loss of dye or pigment during the known solvent-removal procedures.
In recent years, a varnish removal formula was developed by Richard Wolbers at the University of Delaware for painting conservation. It has been used by painting conservators for several years, and has been recently adapted by the author for use in the conservation of varnished paper artifacts.
Varnishes can be divided into two main categories, solvent type and oil resin type. Natural resin varnishes such as mastic, dammar, and shellac belong to the solvent type, which consists of a volatile solvent and a nonvolatile substance. Once applied to an artifact, the solvent dries and the non-solvent substance forms a film coating to protect the artifact against deterioration and to improve its appearance. Indeed, properly applied varnish provides a barrier and protects an artifact from dust and atmospheric pollution, at the same time reflecting ultraviolet light and intensifying color.
Mastic and dammar are natural plant resins obtained as exudates from trees. They are readily oxidized as typical triterpenoid resins, part of a large class of compounds containing 30 C (carbon) atoms derived from the hydrocarbons C30H50. Because of their solvent solubility and their ability to cause less yellowing over time, these triterpenoid resins are superior to conifers and copals as picture varnishes.
Mastic resin is exuded from Pistacia Lentiscus tree, which grows along the Mediterranean coasts, especially on the island of Chios near Turkey. Its use in art has been traced to the first millenium. Although the formed film becomes brittle in a short period of time, it is less likely to wrinkle than dammar. Mastic resin can be completely dissolved in polar solvents and partially dissolved in all organic solvents.
After centuries of use, mastic was superseded by dammar resin as a varnish for paintings in the early 19th century. Dammar is a general name for a group of resins exuded by the Dipterocarpaceae family of trees, primarily indigenous to eastern India, Indonesia, and Thailand. in its unprocessed solid form, dammar is easily scratched and has little flexibility. in spite of these disadvantages, it was widely used because it can be dissolved in a broader range of solvents and because it becomes brittle over a longer period of time.
The third natural resin, shellac or gum-lac, has been used in India since ancient times. Lac is a resinous substance formed on the Butea Frondosa tree by a variety of scale insect, Tachardia Lacca. When the secretion contacts air, it solidifies, forming a protective "scale" or "cell" around the insect. They are then collected as raw "sticklac", washed melted, filtered, and rehardened into shellac. Its chemistry is complicated; however, commercially produced lac, such as French polish, contains mostly Aleuric acid, prepared by extracting shellac from alkali. Exported from India since the 10th century, shellac was used in the production of red dyestuff. in the 19th century, its use was expanded for paper sizing, the production of lithographic ink, and other purposes. the soft, shiny effect of the lac coating was explored by artists of the period to create special effects and eventually, it became a paint medium in its own right.
Mastic, dammar, and shellac have all been used as varnishes to protect paintings from deterioration. However, over time, the varnish coating itself undergoes physical and chemical changes, becoming hard and brittle, and developing discoloration due to oxidation and other complex reactions enhanced by heat and light.
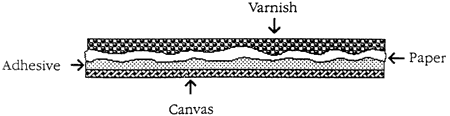
It the artifact is a painting, the deterioration of the varnish affects the paint layer underneath less than if the artifact is paper. Varnish applied over a paper artifact penetrates into the porous material, binds with the paint layer and often with the paper itself. When the varnish becomes hard and brittle, it promotes the delamination and the flaking of the medium, actually accelerating the deterioration process. the cracking, chalking, and yellowing of the varnish coating not only detracts from the visual appearance of an artifact, but also exposes it to atmospheric gases and moisture.
Even more complex conservation problems are faced by the conservator in working with paper artifacts (especially maps) lined with textile. Lining an artifact with textile does not prolong its life. in fact, it may have a greater predisposition to deteriorate due to the incompatibility of the various structural layers. If the layers expand and contract differently, the tension created between the layers will be destructive.
Figure 1
Recently, the author treated a series of 19th century maps of Tompkins County and New York State, which are part of the collection owned by Cornell University. One of the maps, an engraving, was published by John Rutherford in 1872. Hand-colored, it depicts images of waterfalls and important buildings in the area. Originally varnished and lined with textile, the map was in a poor condition. After carefully examining and testing the artifact, the author determined that varnish removal was necessary to stabilize the maps' condition. to minimize the stress during the removal procedure, the author decided to try to adapt a relatively new technique by Richard Wolbers for painting conservation.
A varnish removal mixture, consisting of Carbopol 934 dispersed in ethyl alcohol and mixed with Ethomeen C-25 was prepared. Carbopol resins are highly water and polar soluble acrylic polymers, which function as thickeners and stabilizers for suspension or emulsions with the following structure:
Ethomeen C-25—the common name is Ethoxylated (15) cocoalkylamine—has a great variety of surfactant properties, with the following formula:
Preparation of the varnish removal gel:
2 gm of dry Carbopol was sifted into 100 ml of ethanol which was rapidly agitated. Then it was mixed with 10 ml of Ethomeen C-25. a small amount of distilled water was added drop by drop to maintain an appropriate consistency for this particular application.
Brush varnish removal mixture over Japanese tissue
After the gel was prepared, it was brushed over the surface of the map through Japanese tissue and left to set. the alcohol in the gel softened the old film, gradually dissolving the substance, absorbing it and leaving no stains on the paper. After the varnish was removed, the paper artifact was sandwiched between the two sheets of Japanese tissue, Gore-Tex, moistened blotters, and placed under plexiglass. Then the textile secondary support was easily peeled off and the paper artifact was washed on blotters. Water stains that had developed were reduced with a 2.5% aqueous solution of ammonium hydroxide. the artifact was then lined with Japanese paper and starch paste, dried, and encapsulated.
The same technique for varnish removal was used with a mixed media drawing on paper, dated 1729 by an American artist. the drawing was executed with watercolor, pencil and ink. the drawing originally had been attached to paper board and varnished. There were traces of previous repairs with animal adhesive, which had discolored over time. the main problem was the delamination and flaking off of media as well as paper.
The drawing was tested for varnish and color solubility with various solvents, but unfortunately all of them stained the paper and carried the varnish residue even deeper. Varnish removal gel, however, did not leave stains, but dissolved the film very well. the gel was applied on Japanese tissue laid on the surface. Interestingly, during the procedure, the color of the gel changed from transparent to yellow as the gel absorbed the old varnish. the protective Japanese tissue was kept on the surface of the drawing and at the end was peeled off.
When the varnish was removed, the media of the drawing was consolidated by the spray application of an aqueous solution of funori (a seaweed colloid) on the suction table. the tears and breaks were repaired with starch paste and Japanese tissue. Further conservation treatments proceeded as usual.
This varnish removal technique has the following advantages:
- the gel is non-toxic, (however, the conservator should wear gloves when applying the gel, since it might irritate the skin.)
- it is easy to apply,
- it gives the conservator greater control over the procedure,
- the gel does not penetrate deep in a porous material but lies on the surface. which reduces the risk of color loss.
Although the technique has mainly been used in painting conservation, it should be further investigated for use in paper by the conservation scientists. the author believes that the technique has potential for application across a wide spectrum of conservation fields.
Acknowledgements
The author is grateful not only to Richard Wolbers for the development of the new varnish removal formula, and the technique of its application in painting conservation, but also to Stephen Bonadies for sharing the methodology and the chemical compounds, which made it possible to adapt this promising technique to the area of paper conservation.
References
1. Doerner, M. The Materials of the Artist. First Harvest/HBJ, 1984.
2. Feller, R.L., Stolow, N., and Jones, E.H. On Picture Varnishes and Their Solvents. National Gallery of Art, Washington, D.C., 1985.
3. Gettens, J.R., and Stout, G.L. Painting Materials: a Short Encyclopedia. Dover Publications, Inc., New York, 1965.
4) Imms, A.D., and Chatterjee, N.C. On the Structure and Biology of Tachar-dia Lacca Kerr. Calcutta, Superintendent Government Printing, 1915.
5. Masschelein-Kleiner, L. Ancient Binding Media, Varnishes and Adhesives. Rome, ICCROM, 1985.
6. Mayer, R. The Artist's Handbook of Materials and Techniques. New York, Viking Press, 1985.
7. Mills, J.S. and White, R. The Organic Chemistry of Museum Objects. London, Butterworths, 1987.
8. Plenderleith, H.J. and Werner, A.E.A. The Conservation of Antiquities and Works of Art. London, Oxford University Press, 1971.
9. Sharp, D.W.A. The Penguin Dictionary of Chemistry. London, Penguin Books, 1990.
10.Stebbing, E. A Note on the Lac Insect (Tachardia lacca), Its Life History, Propagation and Collection. 2nd ed. Calcutta, Superintendent Government Printing, 1910.
11.Thompson, D.V. The Materials and Techniques of Medieval Painting. New York, New York, Dover Publications, Inc. 1956.
Tatyana PetukhovaPaper Conservator
Cornell University Library
Publication History
Received: Fall 1992
Paper delivered at the Book and Paper specialty group session, AIC 20th Annual Meeting, June 2-7, 1992, Buffalo, NY.
Papers for the specialty group session are selected by committee, based on abstracts and there has been no further peer review. Papers are received by the compiler in the Fall following the meeting and the author is welcome to make revisions, minor or major.