IDENTIFICATION OF THE PRE-COLUMBIAN PIGMENT MAYA BLUE ON WORKS OF ART BY NONINVASIVE UV-VIS AND RAMAN SPECTROSCOPIC TECHNIQUESMARCO LEONA, FRANCESCA CASADIO, MAURO BACCI, & MARCELLO PICOLLO
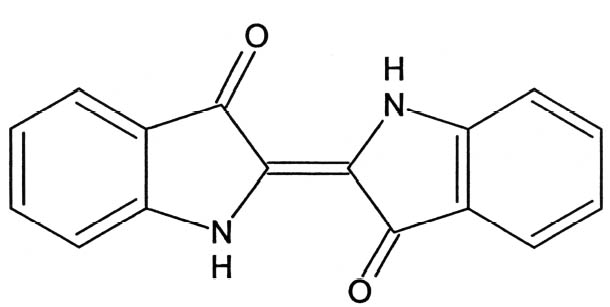
1 INTRODUCTIONSince its rediscovery in 1931 (Merwin 1931), Maya blue has been the object of persistent attempts to unravel its composition and explain its extraordinary stability and colorfastness (Littmann 1980, 1982; Roundhill et al. 1989). While the nature of Maya blue as a complex of indigo and palygorskite had been established by the late 1960s (Gettens 1962; Shepard and Gottlieb 1962; Van Olphen 1966), significant advances in the elucidation of its structure have only recently been made (Polette et al. 2002; Chiari et al. 2003). According to the most recent models, indigo molecules (fig. 1) are hosted in the zeolitic channels of palygorskite crystals (Polette et al. 2002; Chiari et al. 2003), where they engage in hydrogen bonds with the hydroxyl groups of the clay. Palygorskite, whose general formula is (Mg, Al)4Si8 (O, OH, H2O)24�nH2O, is a layer silicate containing ribbon strips of SiO4 tetrahedra and (Mg, Al)O6 octahedra. The ribbons are arranged in such a way as to define channels, which contain water molecules. The structure of palygorskite is too complex to be efficiently depicted here, and the reader should see the above-mentioned references for further information. Early investigations into the nature of Maya blue were based on techniques such as polarized light microscopy (PLM), microchemical tests, determination of elemental composition, or x-ray diffraction (XRD). Of these methods, only the microchemical test with concentrated nitric acid (HNO3) can be described as a specific test for Maya blue, since the persistence of the blue color even after attack with the strong oxidizer is a unique result of the formation of the indigo-palygorskite complex. The other methods
Infrared spectroscopic techniques were later applied to the study of Maya blue with a slightly greater degree of success, due to the possibility of identifying both the inorganic and organic components of the pigment at once, with good sensitivity. However, to date, the only infrared spectra of Maya blue published in the literature are those by Kleber et al. (1967) and by Palacios-Lazcano and Reyes-Valerio (1993). The former, obtained with instrumentation dating back to the late 1960s, are characterized by a rather poor signal-to-noise ratio (S/N), especially in diagnostically significant areas such as the region of wavenumber range 1700–1250 cm-1, and show dominant signals of the inorganic matrix and weak peaks for indigo. The latter, obtained mainly on archaeological samples, contain intense bands due to the clay but also to other contaminants or constituents of the original painting layers such as calcite or gypsum, often completely obscuring the absorption peaks of indigo. More recently, techniques such as transmission electron microscopy (Magaloni et al. 1991; Yacam�n and Serra Puche 1995; Yacam�n et al. 1996; Spoto et al. 2000; Polette et al. 2002), scanning electron microscopy (Polette et al. 1998; Chiari 2001), synchroton x-ray diffraction (Chiari et al. 2003), and luminescence spectroscopy (Aj� et al. 1996; Chiari et al. 1999) have been applied to the study of Maya blue. Electronic and vibrational spectroscopic techniques have so far seen only limited use (Kleber et al. 1967; Palacios-Lazcano and Reyes-Valerio 1993; Grimaldi 2000), in spite of their ability to unequivocally identify the pigment on archaeological samples without the need for sampling and their potential to discriminate Maya blue from simple indigo. Additionally, these techniques are, for different reasons, extremely sensitive to Maya blue. As discussed earlier, detection of indigo in Maya blue by XRD and Fourier transform infrared microspectroscopy (FTIR) is hampered by the small concentration of the dyestuff and by the interfering effect of the clay. The fact that organic colorants, when used as pigments or textile dyes, are always found in extremely low concentrations is an intrinsic problem in the analysis of dyestuffs. To be economically useful, a dye has to have a rather large extinction coefficient; that is, it must impart a strong coloration even when used in small quantities. FTIR and even gas chromatography–mass spectrometry are thus poor techniques for detecting indigo in works of art (Leona 1999), since the analytical signals from indigo are dwarfed by the interfering effect of the other components (binding media, extenders, fragments of the support, or even consolidants, as is often the case with wall paintings and museum objects). UV-visible spectroscopy, compared to the other techniques, uniquely exploits the one physical property of the pigment not shared by extenders and binding media: its electronic spectrum is characterized by absorption in the visible range—color. Raman microspectroscopy can be equally sensitive: Raman peaks are usually sharper than IR bands, and inorganic components may interfere less with the characteristic spectral features of the dye than in FTIR spectroscopy, particularly if the exciting frequency is properly selected (Clark 1995; Cariati and Bruni 2000). In the present study, fiber optics UV-Vis reflectance spectroscopy (FORS) instrumentation and methodologies (Bacci et al. 1992; Bacci and Picollo 1996; Bacci 2000), previously developed for the noninvasive identification of indigo on Japanese paintings of the Edo period (Leona and Winter 2001), were applied in the analysis of an archaeological sample of Maya blue from the excavation of the Templo Mayor in Mexico City (Grimaldi 2000) and, noninvasively, in the examination of a Jaina painted pottery figurine from the collection of the Los Angeles County Museum of Art (LACMA). Raman microspectroscopy (RM), initially explored by Rintoul (Grimaldi 2000) and Guineau (1987), was also successfully employed to identify the pigment in
|