PERMANENCY OF REPROGRAPHIC IMAGES ON POLYESTER FILMHANNA SZCZEPANOWSKA, & WAYNE WILSON
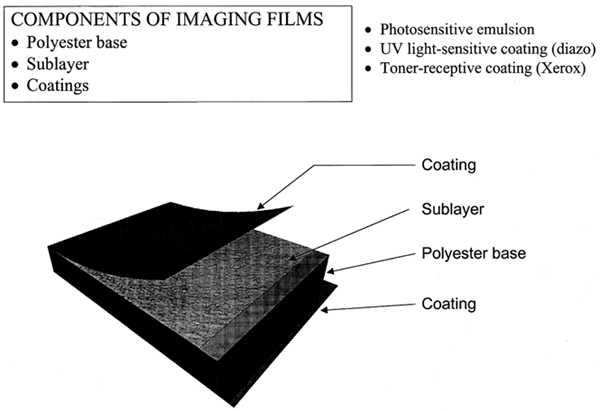
ABSTRACT—ABSTRACT—This project stemmed from a search for archival standards for records made on polyester film. A progressive deterioration was noticed on some records produced during the past 30 years. The damage included discoloration, blistering and flaking emulsion, bleeding images, and images transferred to adjacent pages when records were stored together. To establish handling and storage guidelines that would ensure longevity of records made on polyester film, the project sought a correlation between the ongoing damage and imaging technique and between damage and environmental factors. In an attempt to understand the nature of the damage and the deterioration process, several hundred records from the collection at the Maryland State Archives in Annapolis, Maryland, were examined. Selected samples representing various types of damage and imaging techniques underwent a variety of tests. The effects of exposure to extreme conditions, such as high relative humidity (99%), visible light, and ultraviolet light, are discussed in this article. Although there is substantial literature exploring the history and chemistry of the specific reprographic technologies as well as the characteristics of polyester film, there is no known published information devoted to imaging on polyester film. The experimental work presented in this article is a pilot project, indicating areas that require further in-depth study. TITRE—Permanence des images reprographiques sur film de polyester. R�SUM�—Ce projet est n� d'une recherche sur les normes archivistiques pour les documents sur film de polyester. Une d�t�rioration progressive a �t� not�e sur quelques documents produits pendant les trente derni�res ann�es. On a observ� la d�coloration, le boursouflement et l'�caillement de l'�mulsion, des images qui d�teignent et le transfer d'images sur les pages adjacentes quand les documents ont �t� rang�s ensemble. Afin d'�tablir des directives pour la manipulation et l'entreposage qui assureraient la long�vit� des documents sur film de polyester, on a cherch� une corr�lation entre les dommages observ�s et la technique de capture d'image, ainsi qu'entre les dommages et les conditions environnementales. Afin d'essayer de comprendre la nature des dommages et du processus de d�t�rioration, plusieurs centaines de documents appartenant aux Maryland State Archives (archives de l'�tat du Maryland) � Annapolis, Maryland, ont �t� examin�es. Une s�lection d'�chantillons repr�sentant diverses techniques de formation de l'image et diverses formes de d�t�rioration a �t� soumise � une vari�t� de tests. Les effets de l'exposition aux conditions extr�mes, telles que l'humidit� relative �lev�e (99%) et de grandes quantit�s de lumi�re visible et ultraviolette, sont discut�s dans cet article. Bien qu'il existe une litt�rature consid�rable sur l'histoire et la chimie des technologies reprographiques sp�cifiques, ainsi que sur les caract�ristiques du film de polyester, il semble qu'aucune publication n'ait �t� consacr�e � la formation d'images sur film de polyester. Le travail exp�rimental pr�sent� dans cet article est un projet pilote, indiquant les domaines qui m�ritent une �tude plus appronfondie. TITULO—Permanencia de im�genes reprogr�ficas en pel�culas de poli�ster. RESUMEN—Este proyecto surgi� de una investigaci�n para establecer est�ndares de conservaci�n para documentos realizados sobre pel�culas de poli�ster. Se observ� un deterioro progresivo en algunos documentos producidos durante los �ltimos 30 a�os. El da�o inclu�a alteraci�n del color, ampollamiento y desprendimiento de la emulsi�n, color corrido en algunas im�genes, y transferencia de im�genes a p�ginas adyacentes cuando los documentos se encontraban almacenados juntos. Con el prop�sito de establecer lineamientos para la manipulaci�n y almacenamiento que garanticen la longevidad de los documentos realizados en pel�culas de poli�ster, el proyecto busc� una correlaci�n entre el da�o progresivo y la t�cnica de reproducci�n de la imagen, y entre el da�o y los factores ambientales. Con el fin de entender la naturaleza del da�o y del proceso de deterioro, fueron examinados varios cientos de documentos de la colecci�n de los Archivos del Estado de Maryland en Annapolis, Maryland. Muestras escogidas representativas de varios tipos de da�os y de t�cnicas de reproducci�n de la imagen fueron sometidas a una variedad de pruebas. Los efectos de exposici�n de las muestras a condiciones extremas - tales como humedad relativa alta (99%), luz visible, y luz ultravioleta- son discutidos en este art�culo. A pesar de haber encontrado una cantidad considerable de literatura que explora la historia y qu�mica de las tecnolog�as reprogr�ficas espec�ficas, as� como acerca de las caracter�sticas de pel�culas de poli�ster, no se conocen publicaciones dedicadas a la t�cnica de im�genes sobre pel�culas de poli�ster. El trabajo experimental presentado en este art�culo es un proyecto piloto que se�ala las �reas en las cuales se necesitan estudios m�s profundos en el futuro. 1 1. INTRODUCTIONThe Maryland State Archives is a central repository for state government records of permanent value. The collection of records that was examined for this project encompasses subdivision plats filed at county circuit courts. The records contain information on property ownership, use, and land development. Many are being retrieved as vital support in court disputes, so the legibility and clarity of images is of great importance.1 The archives staff first noticed the image alterations more than a decade ago. However, only recently has a systematic analysis of the collection with respect to damage been undertaken. The first symptom of damage was transfer of images reproduced as xerographic copies on polyester film. These records had severe blocking problems, and recorded information became illegible shortly after they were made. Since then alternative reprographic techniques have been employed. Xerox Corporation researched and experimented with film and coatings that would be more suitable for archival applications (Xerox Corp. 1992). During recent records processing and rehousing, additional surface and image alterations were noticed. The predominant types were discoloration and powdery residue, ink bleeding and image transfer to adjacent pages, and peculiar odors. Several hundred records were examined visually to assess their condition and to select samples for further testing. A database was used to catalog the records, note specific damage, and prepare condition reports. The database also facilitated retrieval of damaged records. Records that were issued as duplicates and were scheduled for disposal served as a source of samples used in the experimental work. The laboratory work on the film samples was supplemented with information on the reprographic techniques and films obtained in interviews with representatives of companies involved in production of the film, including Agfa Graphic Division of Bayer Corporation, Xerox Corporation, DuPont Films Enterprise, and local companies involved in commercial reprography, including Reprographic Technologies of Maryland, Arkwright of Rhode Island, and Azon Corporation of New York. The Maryland State Archives advocates establishing statewide guidelines for the court clerks as to stability of the records they accept and storage of records. Therefore, this study has far-reaching implications. 2 2. GENERAL STRUCTUREMaking an image on polyester film is a complex process. The film itself, the variety of coatings, and the applied imaging chemistry all play an important role in the successful printing and longevity of an image. The basic structure observed in all samples tested was typical of reproductive archival films (fig. 1). Coatings were present on both sides of the film to improve the functionality of the product. One side of an archival storage film may have two or more coatings, one containing the actual image being stored. Because many of the polymeric binders within which the imaging chemicals are dispersed do not adhere well to bare polyester film, a sublayer coating may be used to improve the anchorage of the image to the film.
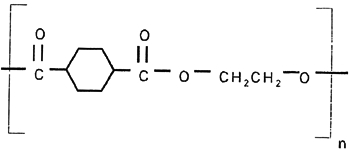
The imaged side of the tested samples was coated with a photoreactive layer, a UV light-sensitive layer, or a toner-receptive layer. In most cases the reverse side was coated with a frost or clear coating and/or antistatic material. The frost coating serves two purposes: to improve the legibility of the image and to provide slip characteristics so the drawing does not block (stick to) the neighboring sheet when stacked. 3 3. POLYESTER FILM AS SUPPORT FOR IMAGING3.1 3.1 GENERIC FILM STRUCTURE AND CHARACTERISTICSThere are approximately 220 polyester film manufacturing facilities around the world. The largest producers are DuPont, Mitsubishi, Toray, SKC, Teijin, Chiel, AGFA, Kodak, and 3M. Dimethyl terephthalate (DMT) and ethylene glycol are the primary chemicals used to produce commercial polyester film, for example, Mylar or Melinex (polyethylene terephthalate [PET]) (fig. 2). DMT can be replaced by terephthalic acid (TPA), which changes the production cycle slightly. DMT is used in a liquid form and TPA in a solid form. The primary chemicals undergo a complex process, which includes the following steps: (1) production of the monomer by combining ethylene glycol and terephthalic acid (or DMT); (2) addition of slip additives; (3) polymerization and removal of “excess” ethylene glycol; (4) filtration; and (5) extrusion. The surface characteristics of the polyester film that affect its durability as a support for reprographic purposes and contribute to image quality and longevity are decided fairly early in the production cycle.
The first decisive step is the successful chemical reaction of the raw materials. The second crucial step is the introduction of the slip additives prior to polymerization. The third step is the polymerization of the monomer. During polymerization, some of the ethylene glycol is freed by the chemical reaction, and this “excess” ethylene glycol must be removed for polymerization to proceed. Once the polymer has reached the desired molecular weight, it is then extruded into a cast sheet. After extrusion, during stretching of the film, crystallization of the polymer begins and removal of “excess” ethylene glycol takes place. Crystal growth occurs in the heat-setting process (DuPont Films Enterprise 1996). Uncoated film is stable in ambient temperatures. In many applications it does not change, even when exposed to a temperature of up to 150�C (302�F).2 The film is inert to most organic solvents at ambient temperature but is hydrolyzed by strong acids and bases and, at elevated temperatures (above 100�C), by moisture (DuPont Mylar 1996). Mylar is resistant to attack by fungi and bacteria. Samples buried in soil for 36 months remained unchanged (DuPont Mylar 1995). The test report of the material by DuPont Polyester Films provided interesting information regarding the stability of polyester film. One study focused on the effect of sunlight and weather on the physical properties of Mylar polyester film, using both outdoor exposure tests and laboratory instrumental methods. In most applications, polyester film is presumed to have been embrittled when it retains less than 20% of its original tensile elongation. The test conditions for this study provided the maximum possible exposure of the film to ambient light and weather. While such conditions are not normally encountered in typical applications, they do help to establish the minimum performance one might expect. According to this report, polyester film did not indicate any degradation when used or stored indoors under ambient conditions over five years. Film used as book cover material was unchanged in mechanical strength after six years. However, there is no way to calculate accurately the indoor longevity of Mylar. The report also cites the effects of ultraviolet light on polyester film. The degradation is rapid where the sun reaches the film. The film surface is altered by the UV light, and the depth of the degraded layer increases with exposure time. For this reason, heavier, thicker gauges of film last longer. Film cracks when the exposed side is bent. 3.2 3.2 SURFACE COATINGSThe extreme chemical inertness of the polyester film base makes adhesion of a coating much more difficult than with paper or other porous surfaces. The film used in reprographic technologies is often coated on both sides. The image side may be coated with photoreactive materials, usually involving silver or diazo compounds, or with electrocharged coatings as used in Xerox processes. The most typical application processes for frosted and photosensitive coatings involve organic solvents. The purpose of the solvent is to swell the surface of the material to be sensitized (as in diazotype) so that the sensitizer can penetrate through part of its thickness (Kosar 1965). Most materials applied to polyester film are held to the film with polymeric binders. Diazo technology, for example, employs a vinyl acetate/crotonic acid copolymer (Kosar 1965) and butadiene acrylonitrile or butadiene styrene copolymer. Polyester film is pretreated early in the manufacturing process for use in reprographic technologies. Historically, the surface was mechanically “roughened” prior to chemical pretreatment to create a stronger bond between the coating and the film (Lubar 1996). Coating of the polyester film is necessary to create an “anchorage” for the embedded image. Successful imaging depends on combining the materials to ensure the correct chemistry among them—strong enough, yet sustaining physical flexing of the support without affecting the image. Each reprographic technology requires a specific type of coating, usually custom-developed in the reprographic plant. Consequently, it is nearly impossible to establish a correlation among the type of damage, type of coating, and reprographic technology. One of the most prevalent types of damage—discoloration—can be attributed to a range of chemicals that were used as a base for imaging and will be discussed later in this article. Products involving xerographic processes and application of toner require different methods of film-base preparation, such as application of a durable coating to bond to the toner. Requirements ensuring stability of the image in Xerox products are discussed in the next section. The application of various overcoats to polyester film, the stability of the imaging technique, and the anchorage of the coatings to the film all affect the durability of the image during use and storage. These features typically are controlled by the manufacturers, who convert polyester film into reprographic materials. Information on coatings, however, is proprietary, for it is the single element that most directly affects image adhesion to the polyester film and hence the commercial success of the supplier. 4 4. IMAGING TECHNOLOGIESThe literature on images and imaging covers a great variety of imaging techniques, but most of it applies to technologies on paper. Images have many applications today. Most imaging technology fits into one of five categories depending on the way the images are created: (1) electronic imaging; (2) photographic silver halide imaging; (3) electrophotographic imaging; (4) chemical imaging; and (5) graphic arts and mechanical technologies. The imaging systems identified in this study are in three of these categories: (1) dry silver (photographic silver halide imaging); (2) xerographic copies (electrophotographic imaging); and (3) diazo (chemical imaging). An evaluation of these imaging systems was based on the microscopic examination of coatings and images on the sample records. A brief description of each technique follows. 4.1 4.1 PHOTOGRAPHIC SILVER HALIDE IMAGING“Dry silver” is a term used commercially by 3M. Since Kodak-Asahi joined 3M, the product has been described as thermally processed silver materials (TPSM). TPSM, both paper and film, are materials that can be exposed to light and processed by the application of heat to obtain an image. The process, substantially dry, takes place in the temperature range 100–250�C (Encyclopedia of Photography 1984). It combines the photosensitivity and speed of silver halide photochemistry with a dry, thermal development derived from thermographic copying technologies. In current practice, the silver halide grains are suspended in a nonaqueous, nongelatin medium with silver developer. Upon exposure to light or electron beams, a latent image forms in the silver halide (Dessauer and Looney 1989). The product of this process is a hard copy of a silver halide photograph characterized by a sharp and clear image. Compared to other imaging processes, these images are the most stable. An example of the dry silver process of photography was found in samples of the DocuMaster system developed by Agfa Division of Bayer Corporation (Afga n.d.), as illustrated in figures 3a and 3b. The imaged film samples were donated for testing and have undergone the same sequence of experimentation as samples from original records. The samples showed no signs of damage or alteration after extended exposure to high levels of humidity. The coating and image were unaffected and were not detached from the film base. A small number of records examined were executed in a technique similar to DocuMaster, producing records that endured extended exposure to high humidity. The Standards Subcommittee IT9-4, which focuses on film stability, was established in 1988 with the purpose of preparing specifications for dry silver film. However, according to the most recent publication (Adelstein 1991), there is no activity in this field, nor is there any current indication of interest.
4.2 4.2 ELECTROPHOTOGRAPHIC IMAGING—XEROXXerography was invented by Chester Carlson (1901-1968) as a nearly real-time, dry-processed, inexpensive office copying system. It is an electrophotographic imaging system. The term “electrophotography” evolved in the 1950s to refer to systems that create images based on electron charges and transfer them onto paper or film. The positive-charge image formed on the photoconductor (toner image) is transferred to the paper or film support by a charging sequence and is thermally fused to the support to make the final image. Figure 4 illustrates the sequence of xerographic image production (Stolka 1989). Toner particles are composed mainly of thermoplastics containing 5–10% of colorant by weight (Scharfe et al. 1989). The function of the thermoplastic is to physically fuse and fix the image on the paper or film by heat, pressure, or a combination of both. Toners are usually fabricated by mixing pigment and polymer followed by melting. The need to employ high temperatures and light in the execution of the Xerox copy requires specific film and coating to withstand demanding conditions. In selecting developer material, a delicate and precise balance of physical, chemical, mechanical, and electrical properties is required, matched to the design characteristics of the machine in which they are used (Xerox Corp. 1992). If this balance is not achieved, the image does not hold to the substrate, as illustrated in figures 5a and 5b.
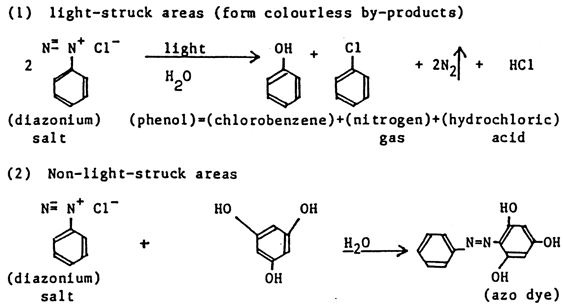
4.3 4.3 CHEMICAL IMAGING TECHNOLOGY—DIAZOMost record samples tested were found to be of the diazo imaging technology. The diazo process was first described by Green, Cross, and Bevan in Great Britain in 1890, but successful commercial products were not marketed until around 1923 (Dessauer and Looney 1989). The large-scale application of diazo printing in imaging technology dates to 1924. It has been used mostly for engineering drawings, color proofs, and microfilm duplicates. Diazo technology produces stable microfilms if one follows the recommended development steps (Kosar 1965). As a reprographic technique on paper, diazo is not a stable technique due to the developing process involved. Images on paper are water-sensitive and fade after prolonged exposure to air and light. The diazotype process gets its name from diazo compounds, which are light-sensitive substances. The diazotype is called “whiteprinting,” since a positive dye image of the original is formed on a white background. This process contrasts with blueprints, which provide a blue-colored negative copy from a positive master. In chemical imaging technologies such as diazotype, the image is created in coatings (applied to paper or film) by the action of light, usually ultraviolet light, or heat. The photochemical reaction takes place in a coating of diazo salts. A diazo print is created by superimposing the original transparent drawing on a sheet with a UV light-sensitive diazo coating, similar to the process of making contact prints in standard photography. The latent image is developed by exposure to ammonia and water vapor. The image is a product of diazo photolysis either in the dry state or in the gaseous phase. The support, either paper or polyester film, is coated with a photosensitive layer containing an aromatic diazonium salt and azo-coupling components. It is exposed to ultraviolet light or blue light. The photochemical sensitivity of the diazo compounds generally used in diazo papers is limited to a narrow spectral region, 375–420 μm. In the irradiated areas, the diazonium ion decomposes by loss of the diazonium group left in the shaded areas and reacts with azo-coupling components, forming azo dyes. Thus a positive image is formed (Kosar 1965; Laurencic 1987). The light to which the diazotype material is exposed must supply sufficient energy to destroy all the diazo compounds in the nonimage areas. The diazonium compounds used in diazotype coatings must meet certain requirements, including sufficient light sensitivity; solubility in water or a common solvent in order to be suitable for sensitizing solutions; and colorless photolysis products to ensure white background. The chemistry of the image formation in the diazo process is illustrated in figure 6.
Microscopically, a diazo image can be recognized by the smooth, clear surface of the photosensitive coating. It does not have the distinctive grain of silver particles found in the silver-based photographic technique. The azo dye images are essentially grainless (fig. 7). (For an example of the overall view of the record, see figure 9a.)
The diazo systems have the advantage of being inexpensive and easy to develop into the final image. The resolving power is about 10 times better than that of silver images. However, the light sensitivity of diazo systems is lower than that of silver photography by a factor of 10 to 100 (Zollinger 1994). According to the findings of standards organizations, diazo images are not considered suitable as “archival” media because they are dye-based. Specifications limits are given in ISO 8225 and IT9.5 for long- and medium-term films (Adelstein 1991). 5 5. DETERIORATION OF RECORDS ON POLYESTER FILM5.1 5.1 TYPES OF DAMAGEThe examined records exhibited various changes that resulted in information loss and/or total record destruction. Some of these changes were caused by poor archival practices, while others resulted from inherent qualities of the materials used for recording. It was observed that older records produced more pronounced discoloration, often combined with emission of peculiar soaplike odors. The following categories of deterioration were noted: discoloration; residue and odors; blistering and separation of emulsion; and image transfer and ink smudges. 5.1.1 5.1.1 DiscolorationDiscoloration was the prevailing type of damage. All other types occurred on a much smaller number of the records. The records examined were unevenly discolored overall. However, there is no apparent correlation between the type of imaging and the discoloration of the records. There was some correlation between the discoloration and the age of records. Older records, created in the mid-1960s, were generally more severely discolored, and residues and odors were more prominent. The degree of discoloration in many cases was more intense in the center than on the margins, excluding such factors as oxidation (figs. 8a–8b).
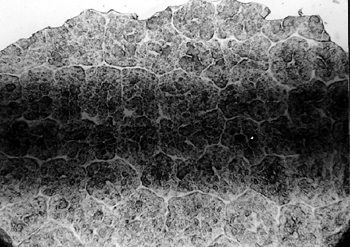
Most of the discoloration was associated with diazotype processing. Discoloration is a result either of slow oxidation of the coupler and the photolysis of the diazo compounds or of the presence of acids. The o-hydroxycarboxylic acids added to the coating (sensitizing solution) to prevent its premature coupling can cause background (nonimage area) discoloration of the prints if used in excess (Kosar 1965). 5.1.2 5.1.2 Residue and OdorsThe formation of a powdery residue was noted mostly on older records (at least 20 years old). In many cases, the residue was accompanied by a distinctive soaplike odor. Microscopic examination did not reveal any particular type of damage or any characteristic features that could be attributed to or connected with the residue formation. The pattern of the residue suggested that it might have been formed after a brief exposure to high humidity, resulting in the breakup of the coating layer. If such exposure occurred, it was not indicated by the microscopic examination. 5.1.3 5.1.3 Blistering and Separation of EmulsionThe separation of the emulsion was dramatically evident on an older record, created in 1974. Microscopic examination revealed that the emulsion was poorly manufactured, resulting in the formation of “alligator skin” (figs. 9a–9d). Interestingly enough, the emulsion did not separate from the polyester film in the expected pattern, following the weak areas of the emulsion. Instead it separated in flakes, revealing a clear layer underneath. The other side of the film was smooth and shiny, indicating the photosensitive layer of the diazo type reproduction (see fig. 7).
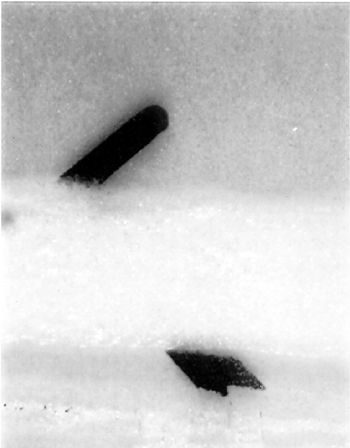
5.1.4 5.1.4 Image Transfer and Ink SmudgesImage transfer is a classic example of the effect of poor storage on archival material. This type of damage was directly related to the type of storage enclosure in which the film was placed (fig. 10a). The enclosure was made of plasticized polyvinyl chloride, which released the plasticizer di-octal-phthalate (DOP). DOP accumulates on both sides, on the surface, and dissolves ink and coatings, resulting in image transfer. The process irreversibly damages the records (fig. 10b).
Another known reason for image transfer and smudges is interaction of organic solvents with toner in xerographic copies. As mentioned earlier, solvents are traditionally used in xerographic processes to ensure adhesion of coatings to the polyester film base. The residual solvents in the coatings interact with toners, resulting in softening of the ink used in creation of an image. Xerox Corporation experimented with aqueous-based coatings to eliminate the problem of solvent-toner interaction. However, the experimental film, aqueous double matte film, never reached the market. 6 6. LABORATORY TESTING AND EXAMINATION METHODOLOGYThe examination of samples of the records was conducted in three stages.
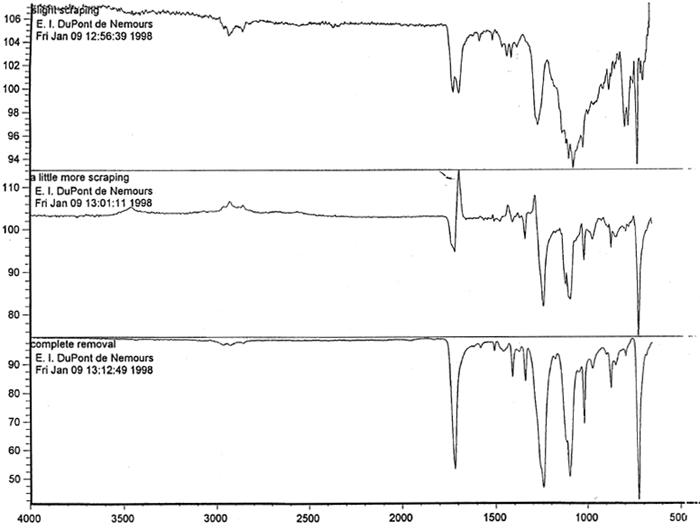
Microscopic examination and analysis of exposed samples. In one set of tests, the methodology was designed to replicate a realistic situation in which the records might be housed. In another set of tests, the methodology replicated exposure to the high humidity level characteristic of extreme environmental conditions. High humidity occurs during natural disasters, such as flooding or fire recovery. The methodology was designed to reveal what alterations occur to records during their exposure to high humidity and how much time passes before the alterations are noticeable. Other methods were based on traditional film-testing standards, such as ANSI standards (ANSI 1989; 1992), and adapted from the film-manufacturing industry. Microscopic analysis of the support, polyester film, coatings, and images and their structure was carried out at 400x. To confirm that the damage was associated with the coating layers rather than with the film itself, the layers were sequentially removed from both sides of the film. The purpose of the testing was threefold: (1) to determine if the film base was indeed polyester; (2) to establish a relationship between the imaging technology and the type of damage; and (3) to determine the effect of extreme environmental conditions on coatings and film. Test samples approximately 0.5 cm wide and 12 cm long were removed from the margins and central portions of the records designated for disposal. The samples were selected in such a way as to represent various types of damage and diversity within each type. For example, the discoloration on the margins and in the centers varied in color and intensity. 6.1 6.1 FILM BASE ANALYSISTo determine if all the tested samples were indeed produced on polyester-base film, several tests were performed. The film samples were prepared for examination by gradual removal of emulsions and backing layers, mechanically and with solvents, to reveal the film base. The analysis was carried out at DuPont Testing Laboratory, Florence, South Carolina, and the Johns Hopkins University Instrumentation Laboratory, Baltimore, Maryland. The base film was analyzed using a Nanometrics NanoSpec 20IR microspectrophotometer (DuPont) and a Fourier transform infrared spectrometer (FTIR), Perkin-Elmer 1600 (JHU), to identify the polymer type. As illustrated in figure 11, the resulting spectra for the cleaned (without coatings) base film were compared with a reference infrared spectrum for a known sample of polyester film. Four strong absorption peaks marked on the spectrum (see fig. 12) are the characteristic peaks for the PET (polyester film) sample. The same characteristics were found in all samples examined, confirming that all tested records were created on polyester film.
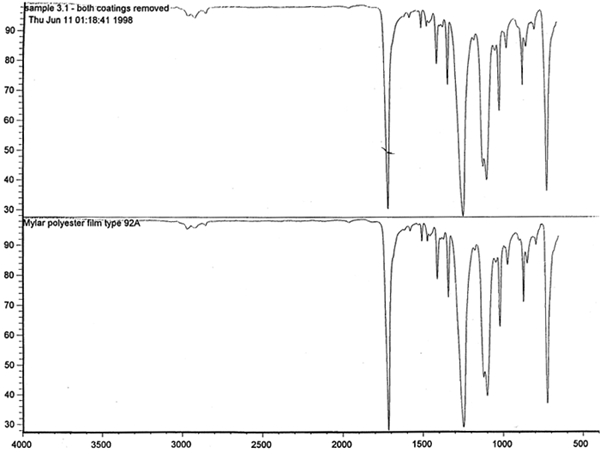
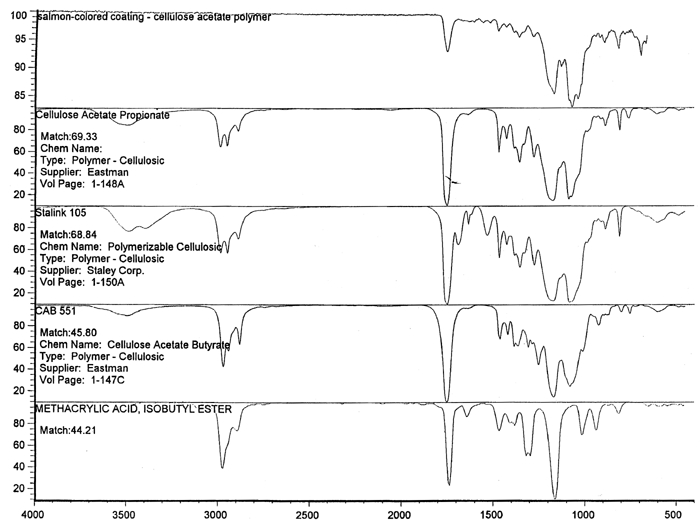
Spectral analysis of film samples collected with FTIR illustrate the reading of the Mylar spectrum while the coating is being gradually removed (see fig. 11). Specific details are obscured when the intact film is examined; a gradual removal of the coating from one surface allows the nature of the base film to emerge. With the complete removal of the coating, the characteristics of the infrared spectrum for polyethylene terephthalate may be seen. The characteristic peaks for polyethylene terephthalate are a single strong absorption peak at 1,715 cm−1 and 1,247 cm−1 and double strong absorption peaks at 1,122 cm−1 and 1,099 cm−1. Figure 12 is a spectrum of a known film sample (a common grade of Mylar polyester film) provided for comparison with the sample film where both coatings were removed. Clearly, both films are made of the same material. 6.2 6.2 COATING ANALYSISTwo samples of the records that varied most dramatically in discoloration (brown and purple) were examined to determine which part of the structure was responsible for discoloration: film or coatings. The coatings were removed one at a time from each side and examined at 10x. Once all coatings were removed, the base film had no discernible color, indicating that the discoloration originated in the coatings. To establish which of the two coatings, imaged or reverse, was discolored, additional samples of 15 records were tested. Each side was examined separately after the coating was removed. In all cases, the discoloration was restricted to the image side. To determine which component of the imaged film was responsible for the discoloration, coatings were removed from each side separately. Coatings that were affected by water typically were gelatins. Coatings that dissolved in other mild solvents usually indicated that other organic binders (such as acrylics, polyurethanes, polyesters, and others) were used in the process of film preparation. The portions of the sample not subjected to solvents for the removal of the coatings were analyzed instrumentally, both with FTIR (to identify organic compounds) and with scanning electron microscopy (SEM)/x-ray multichannel scaling (for elemental analysis). The imaging layer of representative samples was examined with SEM, using JOEL model JSM-T300. The spectral analysis was carried out with a Nicolet Nic-Plan infrared microscope, attached to a Magna-IR 560 spectrophotometer, at the DuPont Laboratory. The elements found in tested samples were (1) silver and chlorine, which might indicate silver-base chemistry but does not preclude diazo technology; (2) titanium, which most likely comes from frosting buildup; and (3) silicon, most likely a component of a slip additive and/or frosting. Figure 13 illustrates an FTIR reflectance spectrum for a typical binder system found in the image-sided coating on an archival record that had discolored. While it is not possible to identify the exact resin used, it is very similar to several cellulosic resins found in the OMNIC reference library. Cellulosic resins are commonly used in imaging applications. (The OMNIC reference library is a collection of optional software including Aldrich Solvents Spectral Library, Polymer Additives and Plasticizer Spectral Library, and Coatings Technology Library. All are used as a resource in comparative studies of spectra of various chemicals.)
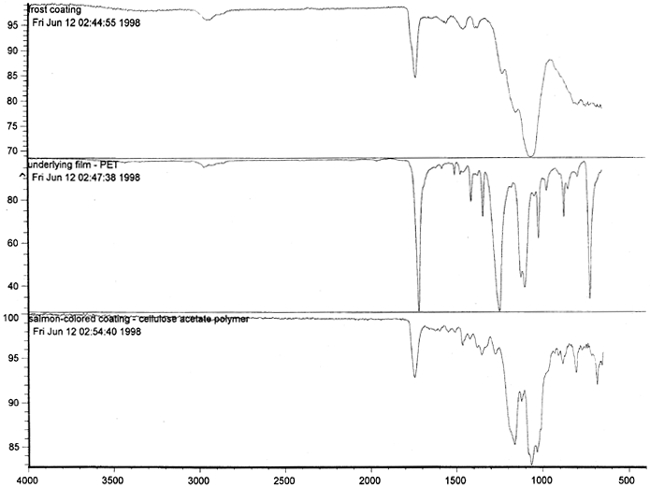
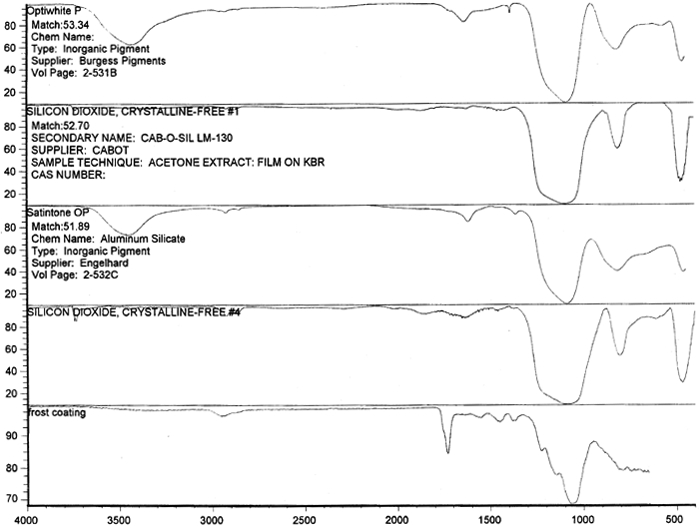
Figure 14 shows three FTIR spectra for the three components found in the salmon-colored sample. These components are the frost coating, the underlying film, and the salmon-colored imaged coating. Comparison of the frosted coating to reference spectra permits the identification of the filler as some form of silica, such as silicon dioxide, a major component of sand (fig. 15). The binder used for the imaged coating can be tentatively identified as a cellulosic resin or perhaps an acrylic.
6.3 6.3 EFFECT OF EXPOSURE TO HIGH HUMIDITY (98% RH)Under normal circumstances in a controlled storage environment, the records should never be exposed to severe environmental conditions, such as 98% humidity. Yet, such conditions may occur during shipment or as a result of a fire rescue operation. The samples of 15 records on polyester film representing various types of damage were placed in a glass laboratory jar so that each sample was freely exposed to the surrounding air and separated from contact with other samples. The samples were exposed for 7, 14, and 21 days to 98% relative humidity. The glass container was placed in a dark room at an average ambient room temperature of 24�C. The surface topography of the samples before and after exposure to high RH was examined with a Nikon Optiphot microscope equipped with differential interference optics (DIC) to highlight the surface characteristics. 6.4 6.4 SUMMARY OF RESULTSAt 7 days' exposure, cracking began to show in the coating of discolored samples. After 14 days' exposure, there was severe cracking of discolored coatings. At 21 days' exposure, the severity of cracks did not increase, but the image was affected. It showed characteristics of dissolved ink and smudged when touched. Some of the samples curled and adhered to each other as a result of the antiblocking agent's losing its stability. Attempts to separate the adhered samples resulted in emulsion splitting (fig. 16). The diazo layers cracked in a hairlike (fig. 17) or parallel pattern (fig. 18). The cracking of the gelatin-coated portion radiated from the photosensitive particle centers.
Based on the microscopic examination of the crack pattern, it appeared that in one case the particles that were photosensitive expanded faster than the emulsion, becoming centers of each crack. In another example, the particles changed color and gathered in clusters between parallel hairlike cracks. 6.5 6.5 EXPOSURE TO VISIBLE AMBIENT LIGHT AND UV LIGHTThe samples were exposed to visible ambient light, with moderate exposure to sunlight. The samples also were exposed for two and three weeks continuously to UV light emitted from a Blacklite Blue bulb (F15T8-BLB, Sylvania, 356nm). Microscopic examination of the coatings on 15 samples after each period of exposure did not reveal any appreciable changes. Similarly, a spectral FTIR analysis of the exposed samples did not indicate any changes. Product information on the polyester film indicated that exposure for 500 hours (approximately three weeks) to a UV light renders the product brittle. The exposure level of UV light was compared to the light levels of a bright sun in Arizona. Degradation of the film starts at the surface, and the depth of degradation increases with exposure time. For this reason, heavier gauges of film last longer. 7 7. CONCLUSIONS AND RECOMMENDATIONSBased on the examination and preliminary tests conducted on the samples of records made on polyester film, the following recommendations are suggested:
The evidence presented here is intended to serve as a base for more extensive research on the complex problem of deterioration of images created on polyester film. The authors believe that evaluation of the imaging systems that were encountered in the course of this work and the analysis of deterioration processes will alert the curators and users of the records to the problems of film permanency and the need to adequately care for records on polyester film. ACKNOWLEDGEMENTSThe authors wish to acknowledge the assistance of Charles A. Long, instrumentation specialist, Chemistry Department, Johns Hopkins University, Baltimore, Maryland; and Douglas Nishimura, Image Permanence Institute, Rochester Institute of Technology, for their constructive criticism and suggestions; Susan Page, senior conservator, and Kitty Nicholson, supervisory conservator, National Archives, for sharing information on their drafting film research. The authors are appreciative of the various company officials for answering questions and sharing information on their processes, materials, and research. The reader should be aware of the difficulties, countless hours, and hard work that go into ferreting out such proprietary information. Not every company is generous in its willingness to share information that may be so necessary to the conservator. Thus, the authors are indebted to those who were so willing to share this information. They include Paul Stouffer, government business manager, Imaging Systems, Agfa Division, Bayer Corporation, for valuable information on Agfa processes, films, and imaging systems; Bill Sparwasser and Robert Ford, Reprographic Technologies, Baltimore, Maryland, for assistance with identification of commercial reprographic systems and a comprehensive tour of the plant; Christine Montgomery, president, Annapolis Copy Center, Inc., Annapolis, Maryland, for information on commercial reprographic processes. The authors would also like to acknowledge Rafal Szczepanowski, RIT School of Industrial Design, for computer graphics illustrations; Ann N'Gadi, Research Library, Smithsonian Center for Materials Research and Education, for her patient compliance with numerous literature search requests; and Betty Seifert, chief conservator, Maryland Archeological Conservation Laboratory, Jefferson Patterson Park and Museum, St. Leonard, Maryland, for allowing access to the examination and printing unit of a Nikon/Hitachi digital signal processor; and the Maryland state archivist for enabling presentation of the results of this work at the 1996 AIC Annual Meeting in Norfolk, Virginia. REFERENCESAdelstein, P.1991. International standards on permanence of imaging materials. In Sauvegarde et conservation des photographies, dessins, imprim�s et manuscripts. Paris: Actes des journ�es internationales d'�tude de l'ARSAG. 22–28. Afga Division, Bayer Corporation, Graphic Systems. n.d. Agfa in the drafting office. Material information sheet 343 (EM). ANSI. 1989. Silver-gelatin type: Specifications for stability. IT9.1-1989. American National Standard for Imaging Media (Film). ANSI. 1992. Ammonia-processed diazo films: Specifications for stability. IT9.5-1992. American National Standard for Imaging Media (Film). Dessauer, R., and C. E.Looney. 1989. Low amplification imaging systems. In Imaging processes and materials, ed. J.Sturge, V.Walworth, and A.Shepp. New York: Van Nostrand Reinhold. 263–279. DuPont Films Enterprise. 1996. Personal communication with staff in Florence, S.C., plant.. DuPontMylar. 1995. Mylar product information chemical properties sheet (12/95) 247432A. DuPontMylar. 1996. Product information, physical and thermal properties sheet (2/96) 222367C. Encyclopedia of Photography. 1984. 1st ed. 279–91, 463–64. Kosar, J.1965. Light sensitive systems: Chemistry and application of nonsilver halide photographic processes. New York: John Wiley and Sons. Laurencic, T.1987. Duplicate plans, their manufacture and treatment. ICCM Bulletin13(3&4):139–47. Lubar, M.1996. Personal communication, research director, Azon Corp., Azon City, N.Y.. Scharfe, M., M. D.Pai, and R. J.Gruber. 1989. Electro-photography. In Imaging processes and materials, ed. J.Sturge, V.Walworth, and A.Shepp. New York: Van Nostrand Reinhold. 135. Stolka, M.1989. Hard copy materials. Chemtech (August):487–95. Xerox Corp. 1992. Xerox Engineering Systems 4 mil aqueous double matte film workbook. Zollinger, H.1994. Diazo chemistry. 2 vols. New York: Weinheim. AUTHOR INFORMATIONHANNA SZCZEPANOWSKA is a conservator of art on paper and parchment. Since 1996 she has been a supervisory conservator at the Maryland State Archives, in Annapolis. In 1978, she received her Masters Degree from the University of Nicolaus Coperincus in Torun, Poland, and in 1985, she became an AIC Fellow. As a Fulbright Scholar in 1989 and 1990, she was a conservator and consultant for the collection of the Knights of Malta at the National Library in Valletta, Old Master Drawings in Medina. This fall, she began consultation work for the Archaeological Museum in Cairo. WAYNE WILSON is technical service manager for DuPont Teijin Films, a leading global supplier for biaxially-oriented polyester films. He is a chemical engineer and has 25 years of experience with DuPont and now DuPont Teijin Films, 17 of which has been spent in the production of Mylar, Melinex, Kaladex, and Cronar polyester films. Received for review November 7, 1996. Revised manuscript received July 1, 1998. Accepted for publication December 1998. NOTES1.. Information on the holdings of the Maryland State Archives can be found at www.mdarchives.state.md.us. Reports detailing the condition and damage of records were compiled by Dawn Steely, processing archivist, Maryland State Archives. 2.. Information about service temperature and other properties of the polyester film can be found at www.dupont.com/polyester in the product literature, document no. H-37232-2, p. 5.
 Section Index Section Index |