OBSERVATIONS ON CYCLODODECANE AS A TEMPORARY CONSOLIDANT FOR STONEREN�E STEIN, JOCELYN KIMMEL, MICHELE MARINCOLA, & FRIEDERIKE KLEMM
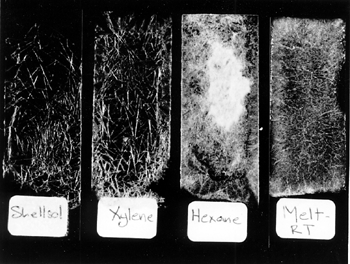
ABSTRACT—ABSTRACT—Cyclododecane (C12H24) is a solid cyclic hydrocarbon that sublimes at room temperature. Cyclododecane has been used increasingly as a temporary consolidant for a variety of weak or friable materials. This study focuses on the physical behavior and working properties of cyclododecane when applied as a solid and in solution to both porous and nonporous substrates. Melted cyclododecane and saturated solutions of the consolidant in hexanes, xylenes, and Shellsol OMS were applied to three different substrates: glass slides, Monks Park limestone, and Ohio Massillian sandstone. Solution mobility, application techniques, and penetration were assessed. Sublimation was monitored in three ways: visually, as a function of weight loss over time, and via gas chromatography-mass spectrometry. This research evaluates the extent of sublimation of cyclododecane from both porous and nonporous substrates and notes the absence of detectable residues. Both substrate porosity and delivery method were found to affect consolidant penetration and rate of sublimation. The practical tests, analyses, and case study indicate the appropriateness and usefulness of cyclododecane for temporary applications on stone substrates. TITRE—Notes sur l'utilisation du cyclodod�cane comme consolidant temporaire pour la pierre. R�SUM�—Le cyclodod�cane (C12H24) est un hydrocarbure cyclique solide qui sublime � la temp�rature ambiante. Le cyclodod�cane est de plus en plus utilis� en tant que consolidant temporaire pour une vari�t� de mat�riaux qui ont perdu leur solidit� ou qui sont devenus friables. Cette �tude se concentre sur les aspects physiques et sur les caract�ristiques de travail du cyclodod�cane losrqu'il est appliqu� en tant que solide ou solution aux substrats poreux et non-poreux. Du cyclodod�cane fondu et des solutions satur�es du consolidant dans l'hexane, le xyl�ne et le Shellsol OMS ont �t� appliqu�s � trois substrats diff�rents: lamelles de verre, pierre calcaire de Monks Park et gr�s Massillian de l'Ohio. La mobilit� des solutions, les techniques d'application et le degr� de p�n�tration ont �t� �valu�s. La sublimation a �t� mesur�e de trois fa�ons: visuellement; en fonction de la perte de poids avec le temps; au moyen d'analyses de chromatographie en phase gazeuse coupl�e � un spectrom�tre de masse. Cette �tude �value le degr� de sublimation du cyclodod�cane des substrats poreux et non-poreux et note l'absence de r�sidus discernables. On a trouv� que la porosit� du substrat et la m�thode d'application affectent le degr� de p�n�tration du consolidant et la vitesse de sublimation. Les essais pratiques, les analyses et les �tudes de cas indiquent que le cyclodod�cane est appropri� et utile pour des applications temporaires sur la pierre. TITULO—Observaciones sobre el ciclododecano como consolidante temporario de piedra. RESUMEN—El ciclododecano (C12H24) es un hidrocarburo c�clico s�lido, que sublima a temperatura ambiente y que est� siendo usado cada vez con mayor frecuencia como consolidante temporario para diversos materiales d�biles o fr�giles. Este estudio se concentra en el comportamiento f�sico y en las propiedades de este compuesto cuando se aplica como s�lido o en soluci�n a sustratos porosos y no porosos. Se aplic� ciclododecano fundido y en soluci�n saturada con tres solventes: hexano, xileno y S hellsol OMS. Se ensayaron tres sustratos distintos: portaobjetos de vidrio, caliza de Monks Park y arenisca Massillian de Ohio. En cada caso se evaluaron la movilidad de la soluci�n, la t�cnica de aplicaci�n y la penetraci�n. La sublimaci�n se control� de tres maneras: visualmente, como p�rdida de peso en funci�n del tiempo y por medio de cromatograf�a gaseosa combinada con espectrometr�a de masas (CG-EM). Este trabajo eval�a el grado de sublimaci�n cuando el compuesto se aplica sobre sustratos porosos o no porosos y la presencia de residuos detectables. Se encontr� que tanto la porosidad del sustrato como el m�todo de aplicaci�n afectan la penetraci�n del consolidante y la velocidad de sublimaci�n. Los pruebas de laboratorio, los an�lisis y los estudios de caso indican que el ciclododecano es un consolidante apropiado y �til para su aplicaci�n temporaria sobre sustratos de piedra. 1 1. INTRODUCTIONCyclododecane has been used with increasing frequency since 1995, when it was proposed for use in conservation by Hans Michael Hangleiter, Elizabeth J�gers, and Erhardt J�gers. Cyclododecane has been used in Central Europe, particularly in Germany, as a temporary treatment for weak or friable materials, including textiles, paintings, paper, architectural elements, painted wood, and various objects. Because cyclododecane sublimes at room temperature with continued air exposure, it has been used in short-term applications, functioning as an adhesive, release agent, barrier layer, or consolidant. Sublimation negates the need for later removal of the adhesive, and temporary treatment with cyclododecane should not interfere with subsequent study, analysis, or treatment. The hydrophobic solid has been applied as a barrier layer over water-sensitive design materials on textiles, paper, and wall paintings to protect them during aqueous cleaning. Gudrun Hiby (1997) illustrated the use of cyclododecane as a local barrier over water-soluble dyes on textiles. Hangleiter (1998b) described the application of cyclododecane over water-sensitive pigments and media in medieval wall paintings to permit aqueous cleaning of the surrounding plaster. Irene Br�ckle et al. (1999) and Cornelia Bandow (1999) tested cyclododecane as a barrier over water-sensitive media during aqueous cleaning of paper. Hiby (1997) also employed cyclododecane in the cleaning of soiled glue-bound paint layers, which were rendered more nonpolar by the consolidant and were therefore not affected by a moderately polar cleaning solvent. Cyclododecane has been used as a facing adhesive or temporary consolidant to stabilize fragile paintings, architectural plasters, and stone objects in preparation for handling or transport (Hangleiter et al. 1995; Hiby 1997). Hiby (1999) tested the use of cyclododecane as a temporary consolidant during transport, using test samples of paintings and underbound polychrome wood sculpture. Cyclododecane has been used to facilitate the excavation of archaeological materials, permitting permanent consolidation to take place away from site in a controlled laboratory environment. Gary McGowan (1999) applied cyclododecane to unbound earth sculptures and then excavated and transported the stabilized sculptures to a laboratory where treatment proceeded after sublimation of the temporary consolidant. Cyclododecane has been tested on low-fire unglazed ceramics to limit the penetration of release agents applied during mold-making and has also been suggested as a barrier layer on painted or porous substrates to prevent retention of fill compounds on surfaces surrounding losses (Br�ckle et al. 1999). 2 2. PHYSICAL AND WORKING PROPERTIESCyclododecane (C12H24) is a translucent, waxy hydrocarbon solid, with a melting range between 58� and 61�C. The 12-carbon molecule is cyclic and unbranched. With continued exposure to air, the solid will completely disappear through sublimation, in a manner similar to camphor. Cyclododecane is readily soluble in nonpolar and aromatic solvents and is poorly soluble in polar solvents. It is insoluble in water and therefore exhibits strong hydrophobicity. Cyclododecane is commercially manufactured as a sealing wax. Br�ckle et al. (1999) reported that it is used as an additive in fragrances and synthetic waxes. Cyclododecane also serves as an auxiliary binder in the injection molding of ceramics and metal powders, improving casting accuracy and efficiency by promoting the removal of binders through sublimation (Hanamura and Katuyoshi 1988; Okamoto 1990). The Material Safety Data Sheet lists low toxicity and gives no restrictions on handling. Cyclododecane is not reported by the supplier, Kremer Pigments, to contain either additives or impurities. Cyclododecane can be heated and applied as a melt, using a brush or heated spatula. Sticks of cyclododecane can be molded to fit into a hot-glue gun or other applicator with a warming chamber. McGowan (1999) described the use of a tool with a heated chamber and Teflon roller. Alternatively, cyclododecane may be dissolved in nonpolar or aromatic solvents and introduced by spray, brush, or injection. The concentration of saturated solutions varies with individual solvents; thus the amount of consolidant delivered by a given volume of saturated solution will vary with solvent choice. Saturated solvent solutions exhibit very low viscosity, and their mobility promotes ease of application by brush, squeeze bottle, sprayer, or syringe, although fast-evaporating solvents may quickly result in clogged needles or nozzles. In order to observe coatings formed by various cyclododecane delivery systems, glass slides were brushed with cyclododecane applied as a melt and as saturated solutions in xylenes, Shellsol OMS, and hexanes. Shellsol OMS, formerly called Sol 71 and Odorless Mineral Spirits, is produced by Shell Chemicals and is described in the manufacturer's literature as a mixture of predominantly branched-chain, saturated hydrocarbons (C3-C12) with less than 0.1% aromatics. These three solvents were selected for evaluation because they provide a range of solubility parameters and evaporation rates. Saturated solutions of cyclododecane were prepared by continually adding known quantities of the consolidant to a known quantity of solvent until no further dissolution occurred and small amounts of solid cyclododecane remained visible in the solution. A saturated solution in Shellsol OMS was found to be approximately 80% wt/wt, while a saturated solution in xylenes was approximately 120% wt/wt and in hexanes was 140% wt/wt. Glass slides were then painted with each saturated solution and with melted cyclododecane. Upon evaporation of the solvents and upon cooling of the melted consolidant, crystal-like structures of cyclododecane formed on the slides. As a wax, cyclododecane is not a fully crystalline solid. The size and pattern of cyclododecane crystal-like structures differ with delivery method (fig. 1). A closely interwoven network of relatively large acicular crystal-like structures formed from the melted cyclododecane. The size and pattern of the coatings formed by the three solvent systems vary with the solvent's evaporation rate. Slides brushed with the Shellsol OMS solution showed, comparatively, a more open network, while those brushed with the faster-evaporating solutions produced tighter networks of smaller crystal-like structures. The consolidant delivery method may, therefore, be modified depending on the type of coating or penetration (discussed below) necessitated by treatment circumstances.
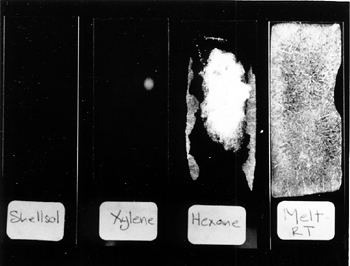
While the glass slides provided a suitable nonporous substrate for assessing superficial coatings of cyclododecane, different methods are necessary for examining the behavior of the consolidant within porous structures. Nicole Riedl and Georg Hilbert (1998) used cryo-scanning electron microscopy (cryo-SEM) analysis to examine porous plaster samples treated with cyclododecane. For this research, melted cyclododecane was applied to plaster samples that had been heated under an infrared lamp for one hour, and a saturated solution of cyclododecane in petroleum ether was brushed on unheated plaster samples. Riedl and Hilbert found that no crystalline structures formed within the substrate pores; instead, a smooth homogeneous film was observed coating the plaster particles of samples treated with both melted and solvent deliveries. These results suggest that there is no probable risk of damage due to pressure exerted on pore walls by crystal growth. The effect on the plaster as the subliming cyclododecane leaves the pores is not yet fully understood. Sublimation can be hastened by increased ventilation or by increased temperature, supplied by a hot-air gun, hair dryer, oven, or heat lamp. Conversely, sublimation can be delayed by sealing the object in a closed environment. Delayed sublimation is demonstrated by the following comparison of two sets of glass slides that were coated with the various consolidant solutions described above. One set of treated slides was exposed to room conditions (approximately 22�C and 50% RH), and the second set was placed into plastic Petri dishes with loose-fitting covers. The Petri dishes were stacked and sealed in a zip-close polyethylene bag. This enclosure, although not airtight, was selected because it replicates the sort of covering that could be used during practical treatment. Within four days, the cyclododecane had sublimed from the unwrapped set of glass slides. The denser coating layers that resulted from the melt and hexanes deliveries required more time to sublime than the more open network of crystals produced from the slower-evaporating solvent solutions. The area of the cyclododecane coatings was observed to diminish gradually from the edges. No traces or residues were visible on the slides following sublimation. After two months in the box and bag enclosure, crystals of cyclododecane were still present on the second set of glass slides. Sublimation did occur to some extent because the packaging was not airtight (fig. 2).
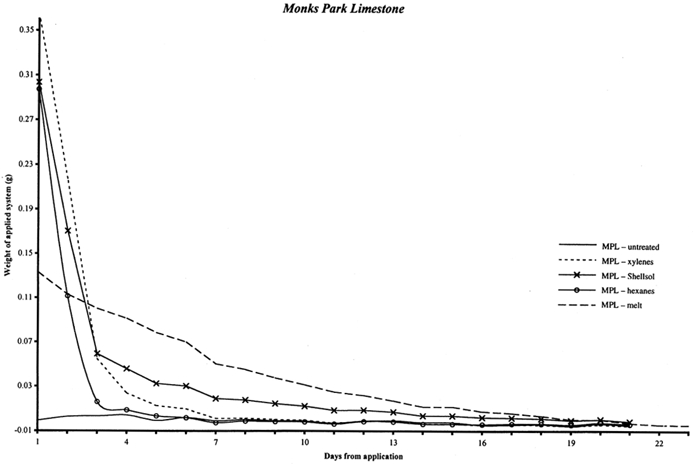
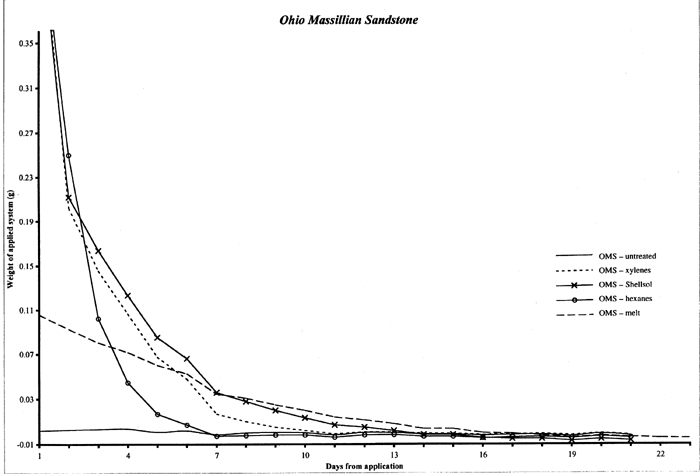
3 3. PENETRATION INTO SANDSTONEIn order to evaluate the penetration of cyclododecane into stone substrates, sandstone samples were treated via various delivery methods. Only sandstone was considered in the initial testing phase for use of cyclododecane on stone substrates; further evaluation of consolidant sublimation included limestone samples as well. Cyclododecane was applied to six cores of Ohio Massillian sandstone, measuring approximately 3 cm in length and 0.5 cm in diameter. Saturated solutions of cyclododecane in hexanes and Shellsol OMS were applied with a brush directly onto one end of the samples; two samples were treated with each solution. The initial depth of penetration, as evidenced by the solvent-saturated surface, was marked with a pencil line immediately following treatment. During the 5–15 minutes following treatment, any further movement of the solution was noted with a second pencil line. Because the 0.5 cm diameter of the core samples provided only a small surface area, melted cyclododecane was painted along one length of two samples. Samples were left overnight to permit solvent evaporation. The untreated ends and lengths of the stone samples were then partially immersed in aqueous dye solutions. The dyes were absorbed by capillary action into the untreated portions of the stone samples, but were repelled by the hydrophobic consolidant, resulting in a visible division between treated and untreated areas. On those sandstone samples treated with melted cyclododecane, the absorbed dye fully saturated the stone and was visible beneath the melted cyclododecane coating, thus illustrating that little or no consolidant penetration had occurred. These stone samples were not warmed prior to treatment, and the consolidant solidified upon contact with the stone surface. Other research has noted varying depths of penetration for melted cyclododecane applied to porous substrates that have been warmed prior to and during consolidant application (Hangleiter 1998b; Riedl and Hilbert 1998; Br�ckle et al. 1999). Penetration of melted cyclododecane up to 1 cm was observed via cryo-SEM by Riedl and Hilbert (1998), while the volatile solvent petroleum ether was observed to carry the consolidant only 2–3 mm into the substrate. On those sandstone samples treated with solvent solutions, the aqueous dye traveled into the unconsolidated stone beyond any notations of secondary penetration and up to the pencil line that marked the initial penetration, at which level the aqueous dye solution was repelled by the hydrophobic consolidant. This absorption of the aqueous dyes suggests a chromatographic effect in which the solvent solutions carried the cyclododecane only as far as initial uptake achieved during application. Any additional penetration beyond the initial uptake therefore appears to reflect the movement of solvent only. Depth of penetration appears variable by solvent evaporation rate, with the slower-evaporating Shellsol OMS solution delivering cyclododecane farther into the sample. Such observations relating to the uptake and penetration of consolidant systems are preliminary, and further testing is needed to more fully evaluate the depths of penetration that can be expected for various consolidant systems applied to different substrates. 4 4. RATE OF SUBLIMATION FROM SANDSTONE AND LIMESTONESublimation of a cyclododecane coating from a nonporous glass substrate was observed to occur within two to four days, depending on the thickness and compactness of the coating. The cyclododecane coatings on the glass slides measured approximately 2 � 4 cm and ranged in thickness approximately 1 to 3 mm. In order to estimate the amount of time necessary for cyclododecane to depart from porous stone substrates, samples of Ohio Massillian sandstone and Monks Park limestone were treated with melted cyclododecane and with saturated solutions. Ohio Massillian is a sandstone building stone commonly used in the United States, while Monks Park is a fossiliferous, calcitic limestone from Bath, England. Both stone types are highly porous sedimentary stones. Mercury porosimetry was performed by Dr. George Wheeler, research chemist at the Metropolitan Museum of Art, on a single sample of each stone. Each stone exhibited a 23% open porosity, and the Monks Park limestone had a greater proportion of larger pores compared to the Ohio Massillian sandstone. Cores measuring approximately 7 cm in length and 0.5 cm in diameter were weighed to 0.00001 g on a mechanical balance. Three replicates per delivery method per stone type were prepared, and three untreated replicates of each stone type were included as controls, totaling 30 samples. Melted cyclododecane was applied with a brush to one end of the samples, and the resulting coatings measured approximately 3 mm in thickness. Approximately 0.5 ml saturated solution of cyclododecane in hexanes, Shellsol OMS, or xylenes was applied to the samples with a syringe. This volume of solution was chosen for experimental consistency, and the complete volume of 0.5 ml of consolidant solution was applied even when the samples appeared to be fully saturated. The fast evaporation rate of the more concentrated saturated hexanes solution resulted in a superficial coating of cyclododecane on the stone. The samples were weighed immediately after treatment and once each day for approximately three weeks until samples had returned to their untreated weights. The accompanying graphs record the gradual sublimation of cyclododecane, as indicated by a loss in weight to zero (fig. 3 for limestone and fig. 4 for sandstone). The weight of the untreated samples was subtracted from the treated weights, so the amount in grams of consolidant introduced by each delivery method is indicated along the y axis. Ripples in the graphed curves, particularly below zero, might be attributed to moisture uptake with fluctuations in the laboratory's relative humidity level, as is further indicated by comparable variations in the curves for the untreated samples. The initial steep declines for the solvent deliveries are interpreted as reflecting the loss of both solvent and superficial cyclododecane. The consolidant sublimes most readily from the sample surfaces, and the greatest loss of cyclododecane is therefore also likely to occur in the first days following treatment. The graphed weights then gradually diminish, presumably as cyclododecane is lost from within the stone substrate. The loss of cyclododecane from within the stone substrate is slower than the loss of consolidant from the surface. Substrate pores do allow sublimation, but they also contain the consolidant and limit its ability to sublime, similar to the effect of enclosing the coated slides in the plastic bag and box packaging. The curves illustrating those samples treated with melted cyclododecane are more linear. The melted consolidant remained on the surface of the sample, without significant penetration into the stone's pores, and is more uniformly lost with continued air exposure.
Table 1 records the number of days required for the stone samples to return to their untreated weights differentiated by application method. Sublimation periods vary in part because each delivery method introduces a different amount of cyclododecane to the stone. Because the greatest amount of cyclododecane was applied by melted delivery, the overall height of this curve is greater, and more time is needed for the consolidant to depart from the substrate. A saturated solution of cyclododecane in hexanes is approximately 140% solids, while a saturated solution in Shellsol OMS is only approximately 80%. Thus a sample treated with a given volume of hexanes solution received more cyclododecane and correspondingly required longer to return to its untreated weight.
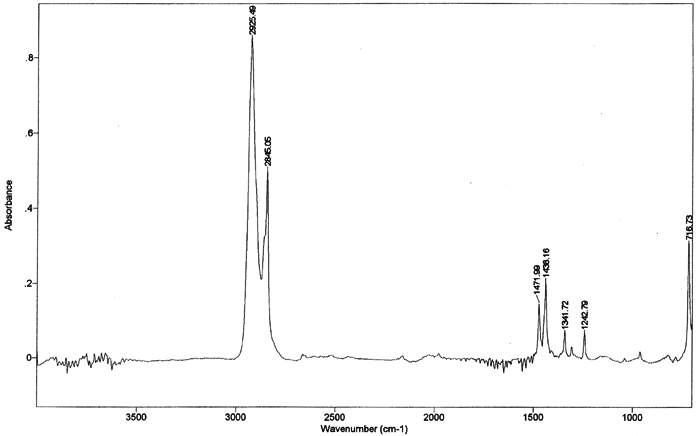
The sublimation times also differ slightly for the two stone types, with limestone samples generally requiring more time to return to their untreated weight. Since experimental conditions were the same for both limestone and sandstone samples, this discrepancy in sublimation rate suggests that inherent stone properties, including porosity, pore size, permeability, and mineralogy, could affect both the uptake of cyclododecane and the rate of consolidant loss. In particular, the Monks Park limestone has a greater proportion of larger pores, which may promote the penetration of cyclododecane. Although, in theory, larger pore size should also allow ease of sublimation, experimental results reflect slightly longer sublimation times for the limestone samples. Further research could elucidate the relative impact of various stone properties on the penetration and subsequent sublimation of cyclododecane. Riedl and Hilbert (1998) report that after nine months of sublimation, cyclododecane was still detectable within the pores of plaster samples treated with melted cyclododecane. Prior to treatment, these plaster samples had been heated for one hour under an infrared lamp at an unspecified temperature, and up to 1 cm of penetration by the melted consolidant was achieved during application. Cyclododecane delivered via saturated solution in petroleum ether sublimed from the plaster samples within five months. Because of both the increased penetration observed on heated samples and the long sublimation times required for these samples, Riedl and Hilbert advise against heating of substrates in preparation for treatment with cyclododecane. Stone samples treated in the present study were not heated prior to treatment, and penetration of the melted consolidant was minimal, with sublimation occurring over a period of days. Discrepancies between results reported by Riedl and Hilbert and those of the present study are largely due to differences in experimental protocol. 5 5. EVALUATION OF PURITY AND EXTENT OF SUBLIMATION VIA FTIR AND GC-MSDue to its ability to sublime at room temperature, cyclododecane will gradually depart from a substrate, and it is not reported to contain any additives that might remain in or on the substrate after sublimation. Purity of the material was examined by analyzing samples from a single can of the consolidant using both Fourier transform infrared spectrometry (FTIR) and gas chromatography-mass spectrometry (GC-MS). As noted above, no residual traces of cyclododecane or of any impurities or additives were visible on the treated glass slides after sublimation. Nor were any residues indicated by the weights of treated stone samples after sublimation of the consolidant. GC-MS was employed as a sensitive technique for the qualitative detection of residues that would be too small to be detected visually or by weight. GC-MS provides highly sensitive yet indirect analysis that enables detection of components on the order of parts per billion. More direct examination of the samples themselves for possible evidence of residues of any sort could be accomplished with cryo-SEM, as utilized by Riedl and Hilbert (1998), or by diffuse reflectance micro-FTIR. The former technique was not available for the present study, and the latter is not as sensitive as GC-MS. A sample of cyclododecane was pressed between a pair of diamond cells and scanned 50 times at a resolution of 4 wavenumbers, using a Bio-Rad FTS 40 spectrometer with UMA 500 microscope. The infrared spectrum of cyclododecane (fig. 5) illustrates a hydrocarbon without carbonyl functionality. Straight-chain paraffins, which might be expected as impurities or additives in cyclododecane, are not reflected in the infrared spectrum. Peaks characteristic of paraffins, around 1150–1130 cm−1 and 1090–1055 cm−1, are absent from the spectrum of cyclododecane.
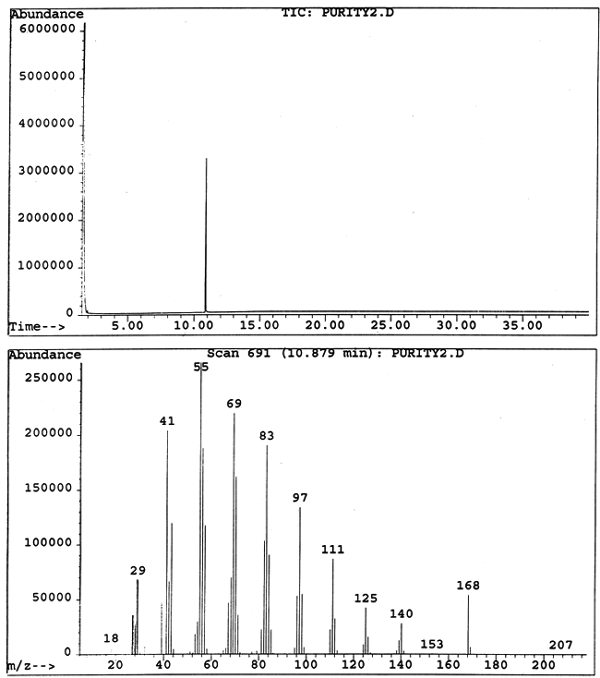
Cyclododecane was then dissolved in ether, and a sample volume of 2 μl was analyzed on a Hewlett Packard GC-MS system, which consists of a 5980 capillary gas chromatograph with split/splitless injection port and 5989B mass selective detector. A Supelco SPB-1 bonded dimethylsiloxane column (30 m, 0.25 mm diameter, 0.25 μm film thickness) was used with helium as the carrier gas at a flow rate of approximately 1 ml/min and a linear velocity of 39.0 cm/sec. The injection port temperature was 250�C, and injections were carried out in split mode with a split ratio of approximately 1:10. The temperature was held for one minute at 45�C, then ramped at 5�/minute to 70�C and then at 30�/minute to 280�C, and held at the final temperature for 27 minutes to give a total run of 40 minutes. MS analysis was carried out in scan mode with a transfer line temperature of 200�C and temperature of the quadrapole ion source at 100�C. This analysis of cyclododecane via GC-MS further demonstrates the material's purity with a single elution of the consolidant at approximately 11 minutes (fig. 6).
Testing for residues in selected stone types was then carried out. The limestone and sandstone cores were treated with melted cyclododecane and with saturated consolidant solutions as described in the preceding section. The samples were weighed before treatment and periodically during sublimation. Once all samples had returned to their untreated weights, approximately four weeks after treatment, consolidant loss was presumed complete. Because GC-MS analysis was in part intended to confirm complete sublimation as indicated by weight, it was necessary to prevent any further sublimation after the four-week weighing period. Several days were required to prepare and analyze each of the 30 samples; therefore, after four weeks the samples were placed in two layers of zip-close polyethylene bags, significantly inhibiting further sublimation as demonstrated in section 2 above. Prior to analysis, each stone sample was crushed with a metal mortar and pestle and transferred to a glass jar with a plastic lid. Then 4 ml of reagent-grade hexanes were added to each jar. The closed jars were gently agitated, and the stone dust was allowed to settle. Because cyclododecane is an unbranched cyclic molecule and unlikely to bond strongly with the substrate, traces of cyclododecane should have been easily extracted from the crushed samples by flushing with hexanes. Five drops of the hexanes solution were transferred with a disposable plastic pipette into 2 ml glass vials with plastic screw-tops. Approximately 1 ml of high-performance liquid chromatography-grade ether was added to each vial, and the vials were gently agitated. Then 1 μl of the ether solution was injected onto the column for GC-MS analysis. Instrumental parameters were as described above, with the exception of the temperature program, which was modified to shorten the time required for each run of analysis. The temperature program for analysis of samples from the treated stones was as follows: the starting temperature of 35�C was held for 1.5 minutes, then ramped at 5�/minute to 70�C and then 30�/minute to 250�C, and held at the final temperature for 1 minute to give a total run of 15 minutes. The sensitivity for this analytical protocol was evaluated on the basis of increasingly dilute solutions of a known concentration of cyclododecane in hexanes and ether. A 1 μl sample of the solution standard containing 1 � 10−8 grams of consolidant yielded an ion abundance of less than 50; thus, it is evident that cyclododecane can be detected well below 1 ppb. Those samples for which GC-MS analysis revealed any trace of cyclododecane were allowed to continue subliming. The jars were opened to evaporate off the hexanes and ether, and the crushed samples were spread onto aluminum foil sheets to promote air contact. These crushed samples were exposed to room temperature and humidity for five additional days and then were returned to the glass jars. The samples were retreated with hexanes and ether and then reanalyzed as described above. Following this extended sublimation period, no further traces of consolidant were detected in any samples via GC-MS. Table 2 shows the results of the GC-MS analysis of the stone samples. After four weeks of sublimation, very small amounts of cyclododecane (ion abundances ranging from <25 to <350) were detected in less than half of the treated samples. The detection of these trace quantities of consolidant confirms the viability of the experimental protocol. Although no consolidant was visible on the stone surfaces and the samples had returned to their untreated weights (recorded to 0.00001 g), traces of cyclododecane remained in 6 out of 12 limestone samples and in 5 out of 12 sandstone samples. Those samples treated with the hexanes system most consistently show small amounts of cyclododecane still present on the stone (5 out of 6 samples). Saturated at approximately 140%, the hexanes solution introduces more cyclododecane per applied volume than the other solvent solutions. Comparatively, the Shellsol OMS solution introduces the least amount of consolidant, and fewer of these samples have retained any detectable trace of cyclododecane after four weeks (only 2 out of 6 samples).
The samples treated with melted cyclododecane received the greatest amount of consolidant, yet 5 out of 6 of these samples show no detectable trace of cyclododecane after four weeks of sublimation. Comparing the results for samples treated with melted cyclododecane with those for the solvent solutions suggests that penetration affects sublimation time. Very little if any penetration occurred with the melted cyclododecane, whereas solvent solutions carried the consolidant well into the substrate. Riedl and Hilbert (1998) instead found that melted applications to heated plaster samples took longer to sublime than saturated solution deliveries of cyclododecane in petroleum ether to unheated samples. Both the quantity of cyclododecane introduced by the melted application and the penetration achieved by heating the plaster samples probably contributed to the longer sublimation times required for the plaster samples. This discrepancy between the sublimation rates of melted cyclododecane on the stone samples and on the plaster samples treated by Riedl and Hilbert further suggests that penetration impacts sublimation time. The present analyses using GC-MS therefore indicate that complete sublimation from within stone pores may actually require more time than can be gauged visually or by weight loss, especially if delivered as a saturated solution. It is, of course, pertinent to subsequent treatment plans to know when all cyclododecane has left the pore structure of the substrate. Correlation between sublimation time and depth of penetration, as determined by delivery method or by substrate porosity, requires further evaluation. For the series of experiments described above, the same volume of consolidant solution was applied to each sample. By instead establishing experimental parameters to introduce a constant quantity in grams of cyclododecane, rather than a controlled volume of consolidant solution, it may be possible to more accurately assess the sublimation rate for particular substrates and for individual delivery methods. 6 6. CASE STUDY: TEMPORARY STABILIZATION OF A DOLOMITIC LIMESTONE COLUMN BASEFollowing testing and evaluation of its working properties, cyclododecane was used as a temporary consolidant to permit the safe transport of a large column base in the collection of The Cloisters, of the Metropolitan Museum of Art in New York City. Part of the 12th-century apse from San Martin at Fuentidue�a, Spain, the base measures 62 cm in width and is carved from highly porous dolomite. The yellow dolomite of the apse from San Martin was characterized by Charola et al. (1986) as 90–94% dolomite and 1–5% calcite, with the balance consisting of quartz, illite, and kaolinite. Its porosity ranges from 19% to 27%, and it exhibits bimodal pore-size distribution. The base required consolidation prior to its transport from the museum during storage renovation. The stone surface is dirty and exhibits granular disintegration, flaking, and scaling. Remnants of polychromy are detached and lifting. Numerous deep and shallow fissures extend across the stone surface. Consolidants such as ethyl silicates and acrylics in various solvents were found to be unsuitable for use on test samples of this same stone (Kimmel and Wheeler 1999). Ethyl silicates did not adequately bridge small fissures in the stone. Paraloid B-72 (acrylic copolymer) caused irreversible staining, as did some test applications of DriFilm 104 (silicone resin in mineral spirits), Conservare H (ethyl silicate and methyl-tri-ethoxysilane formulation), and Conservare OH (ethyl silicate). Temporary consolidation with cyclododecane was chosen to stabilize the stone and prevent damage during transport. Melted cyclododecane was applied by brush to cracks and loose stone fragments, forming a thick opaque coating, which cooled on the surface of the object without appreciable penetration. A hot-air gun was directed at the coated surface in an effort to encourage penetration. Direct heat from a hot-air gun applied at a close working distance was found to provide enough heat to force immediate sublimation. The entire surface, including areas with loose remnants of polychromy, was then coated with a saturated solution of cyclododecane in 3 parts Shellsol OMS to 1 part xylenes by volume. This solution was applied with a wash bottle. Shellsol OMS was chosen as the delivery solvent because of its low toxicity. Xylenes were added to increase the concentration of the saturated solution. As noted above, a saturated Shellsol OMS solution is about 80% cyclododecane, and a saturated solution in xylenes is approximately 120% by weight. Upon evaporation of the solvents, a netlike coating of cyclododecane crystals formed on the stone surface (fig. 7). Immediately following treatment, the base was transported to the new storage location.
The cyclododecane provided adequate consolidation and stabilization of the fragile surface, the loose stone and polychromy fragments, and the cracks. No loss or damage to the stone or the polychromy occurred during the move. No tidelines were formed by the consolidant, despite the layer of surface dirt and the susceptibility of this stone to darkening upon conventional treatment. After approximately 10 weeks, the cyclododecane appeared to have departed, leaving no visible traces of the netlike coating or the melted surface layer (fig. 8).
7 7. CONCLUSIONSThis study describes the physical and working properties of cyclododecane and also addresses the issues of penetration and rate of sublimation from porous stone substrates. Both the amount of cyclododecane delivered to a substrate and the depth to which the consolidant penetrates are variable with application method, whether delivered as a melt or as a saturated solvent solution. The concentration of saturated solutions differs depending on the solvent used, thus varying the amount of consolidant applied to the substrate. The size of consolidant crystals and the compactness of the cyclododecane coating also varies with delivery method. Solvent solutions carried the consolidant into substrate pores, while melted cyclododecane did not readily penetrate unheated porous substrates. Sublimation rates differed for the various delivery methods. More time was needed for the solvent-delivered consolidant to sublime from within a porous substrate than for the melted coating to depart from the surface, regardless of the amount of consolidant applied. It should be noted that, although not part of the present study, the introduction of an additional factor, such as heating of the substrate, may greatly increase both the penetration depth and the subsequent sublimation rate from pore structures. Sublimation rates also differed for the two stone types evaluated, suggesting that substrate properties such as porosity, pore size, permeability, and mineralogy affect consolidant loss. The presence of salts, soiling layers, and materials introduced in previous treatments may also influence the penetration and sublimation rate of cyclododecane applied to porous substrates. Provided adequate time, cyclododecane did depart from stone substrates without leaving residues detectable visually, by weight, or by GC-MS. A more direct method of assessing the extent of sublimation might be desirable when developing protocols for the practical use of cyclododecane, especially when further treatment is needed after sublimation of the temporary consolidant. Determining the amount of time necessary for complete sublimation will require testing for each substrate and delivery method. If treatments with cyclododecane are to be followed by additional treatments, it may be advisable to use application methods that promote only limited penetration, allow plenty of time for sublimation, or encourage sublimation by increasing ambient temperature or air circulation. ACKNOWLEDGEMENTSAll research for this project was conducted at the Sherman Fairchild Center for Objects Conservation at the Metropolitan Museum of Art (MMA). The authors extend their appreciation to the department for the generous use of materials, laboratory space, and analytical equipment. Dr. George Wheeler, MMA, research chemist, offered advice on experimental protocol, commented on drafts, and provided stone samples. Dr. Elisabeth J�gers shared information relating to the ongoing use and testing of cyclododecane at the Fachhochschule K�ln, Germany. Dr. Richard Laursen, MMA, visiting researcher, and Lorna Barnes, MMA, assistant objects conservator, assisted in locating references. REFERENCESBandow, C.1999. Cyclododecan in der Papierrestaurierung. Restauro105:326–29. Br�ckle, I., J.Thornton, K.Nichols, and G.Strickler. 1999. Cyclododecane: Technical note on some uses in paper and objects conservation. Journal of the American Institute for Conservation38(2):162–75. Charola, A. E, L.Lazzarini, G. S.Wheeler, and R. J.Koestler. 1986. The Spanish apse from San Martin de Fuentidue�a at The Cloisters, Metropolitan Museum of Art, New York. Case studies in the conservation of stone and wall paintings: Preprints of the contributions to the Bologna Congress, 21–26 September.London: International Institute of Conservation. 18–21. Hanamura, T., and S.Katuyoshi. 1988. Metal binder and molding composition. European Patent #296552. Assigned to Indemitsu Petrochemical Co., Japan. Hangleiter, H. M1998a. Erfahrungen mit fl�chtigen Bindemittel (Welche Materialien eignen sich und wie wird bei der tempor�ren Hydrophobierung von Wandmalereien vorgegangen?)Restauro104: 314–19. Hangleiter, H. M1998b. Erfahrungen mit fl�chtigen Bindemittel (Teil 2: Vor�bergehende verfestigung sch�tzende oder verd�mmende Versiegelung von Oberfl�chen an Gem�lden, Stein oder Wandmalereien). Restauro104: 468–73. Hangleiter, H. M, E.J�gers, and E.J�gers. 1995. Fl�chtige Bindemittel. Zeitschrift f�r Kunsttechnologie und Konservierung9:385–92. Hiby, G.1997. Das fl�chtige Bindemittel Cyclododekan. Restauro103:96–103. Hiby, G.1999. Cyclododecan als tempor�re Transportsicherung. Restauro105:358–63. Kimmel, J., and G.Wheeler. 1999. The apse from San Martin at Fuentidue�a: Conservation history, survey of conditions, analyses. Metropolitan Museum of Art, New York, N.Y. McGowan, G.1999. Personal communication. Cultural Preservation and Restoration Inc., Hackettstown, N.J. Okamoto, K.1990. Ceramic binder compositions. European Patent #350070. Assigned to Indemitsu Petrochemical Co., Japan. Riedl, N., and G.Hilbert. 1998. Cyclododekan im Putzgef�ge. Restauro104:494–503. FURTHER READINGGeller, B., and G.Hiby. 2000. Fl�chtige Bindemittel in der Papierrestaurierung sowie Gemalde und Skulpturenrestaurierung.Munich: K�lner Beitr�ge zur Restaurierung und Konservierung von Kunst- und Kulturgut, 10. J�gers, E.1996. Partielle Behandlung von textilen Objekten-M�glichkeiten und Grenzen herk�mmlicher und neuer Methoden. Ehemaligentreffen der Abegg-Stiftung Riggisberg1(2):60–71. J�gers, E., and E.J�gers. 1999. Volatile binding media—Useful tools for conservation. In Reversibility—Does it exist?, ed. A.Oddy. London: British Museum Occasional Papers135:37–42. SOURCES OF MATERIALSCyclododecaneKremer Pigment Inc. 228 Elizabeth St. Sources Last Line:New York, N.Y. 10012 Shellsol OMSPectin Chemicals 2 Shell Plaza P.O. Box 1422 Sources Last Line:Houston, Tex. 77252–1422 AUTHOR INFORMATIONREN�E STEIN received her master of science from the Winterthur Museum-University of Delaware Program in Art Conservation with a specialization in objects conservation. She is a contract conservator focusing on the ancient Egyptian collection at the Michael C. Carlos Museum. She was a Mellon Fellow at the Metropolitan Museum of Art and has trained in conservation laboratories in the United States and Italy, including The Cloisters, New York State Bureau of Historic Sites, Colonial Williamsburg Foundation, and National Museum of Natural History. Address: Michael C. Carlos Museum, Emory University, 571 South Kilgo St., Atlanta, Ga. 30322 Jocelyn Kimmel is a conservator of architecture currently based in Portland, Oregon. She has consulted with Myers Conservation, Central Park Conservancy and Art2Facts, and has been employed by Integrated Conservation Resources. In addition, she was a Kress Fellow at The Cloisters, Metropolitan Museum of Art, and was selected by US/ICOMOS to work with the Bundesdenkmalamt in Mauerbach, Austria. She received her master of science in historic preservation from the University of Pennsylvania and has participated in three ICCROM courses. In 2001, she will begin doctoral studies at the Institute of Archaeology, University College, London. Address: 12130 S.W. Morning Hill Dr., Tigard, Ore. 97223 MICHELE MARINCOLA received her M.A. and certificate in conservation from the Institute of Fine Arts, New York University. She is associate conservator for The Cloisters, Metropolitan Museum of Art, where she has worked since 1990 on the examination and treatment of medieval sculpture. Recent publications have included several technical studies of sculpture by Tilman Riemenschneider. She is a professional associate of AIC and a past co-chair of the AIC Objects Specialty Group. Address: The Cloisters, Fort Tryon Park, New York, N.Y. 10040 FRIEDERIKE KLEMM is currently completing her graduate thesis in the conservation of art and cultural objects at the University of Applied Science in Cologne, Germany, focusing on the conservation of stone and mural paintings. She has been involved with stone and wall paintings conservation since 1992 and has been employed by Michael Walta (Bamberg) and Klaus Klarner (Munich), and has completed internships with Metropolitan Museum of Art, The Cloisters, and Preis & Amp/Preis OHG (Regensburg). Address: Semperstrasse 76, 22303 Hamburg, Germany Received for review February 1, 2000. Revised manuscript received June 15, 2000. Accepted for publication July 5, 2000.
 Section Index Section Index |