MOISTURE RELATIONSHIPS OF PHOTOGRAPHIC FILMP.Z. ADELSTEIN, J.-L. BIGOURDAN, & J.M. REILLY
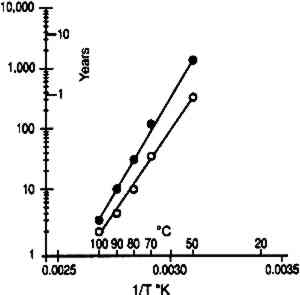
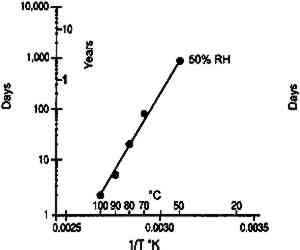
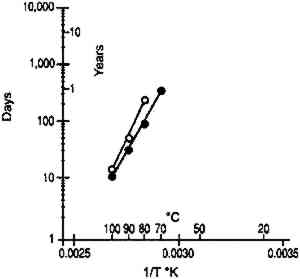
3 DISCUSSION3.1 IMPLICATIONS FOR ACCELERATED INCUBATION TESTSThe standard method used to study the life expectancy of photographic film is to determine the rate of degradation under accelerated temperature/relative humidity conditions. The results obtained at these severe environments can then be extrapolated to practical storage environments using the Arrhenius treatment of the data (Anderson and Kopperl 1993; Reilly 1995). This approach is valid only if the degradation at elevated temperature proceeds by the same mechanism as at the storage temperature. Over the years this Arrhenius procedure has provided very valuable information and insight on the preservation of photographic materials. Two experimental techniques have been used in these studies. The first consists of using ovens that are controlled to a constant temperature and relative humidity. However, as already pointed out, the moisture content of the material changes as the temperature changes. The resulting Arrhenius lines are projections made at constant relative humidity rather than at constant moisture content. The second experimental technique consists of conditioning the samples at a known relative humidity and then sealing them in moisture-proof bags. This procedure ensures that the water content This recommendation is correct for some photographic systems, as illustrated by the example cited above. However, it is not believed to be universally true, particularly when the degradation process is chemical and not physical. The glass transition temperatures obtained from moisture content isolines (table 1) were applied to a recent study on the stability of cellulose ester base photographic film (Adelstein et al. 1992a, 1992b) in which the sealed bag technique was used. Analysis of the cellulose ester base film study was confined to two properties that reflect only characteristics of the emulsion layer. The first is the wet strength of the emulsion that is determined by the weight on a 15 mil. radius sapphire stylus required to rupture the surface when wet. It has been described in the literature as the “emulsion-mushiness test” (Parker and Sugden 1963; ANSI 1992). The second test is the emulsion melting point. It measures the temperature of an aqueous solution in which the emulsion dissolves (ANSI 1964). As the gelatin degrades, both the mushiness and the melting temperature are decreased. Figure 15 shows Arrhenius plots of wet emulsion strength for a motion picture film. Linear plots were obtained when this material was sealed in moisture-proof bags at 21�C when in moisture equilibrium at both 50 and 60% RH. With the exception of the 50�C and 50% RH point, all data were obtained above the glass transition temperature (see table 1). It is estimated that at 50% RH, the glass transition temperature is approximately 52�C. At this temperature, the Arrhenius plot is still linear, suggesting that the glass transition temperature is not a critical parameter for this property. Figure 16 is a similar graph for a motion picture film from another manufacturer. Again there does not appear to be any discontinuity in the Arrhenius plot as the glass transition temperature is approached.
Analysis of the emulsion melting point for the film is given in figure 17. With this property, data are available at 20% RH as well as at higher humidities. It is estimated that 80�C and 20% RH is below but that 90�C and 20% RH, and 100�C and 20% RH, are above the glass transition temperature of gelatin. A linear Arrhenius relationship is still observed.
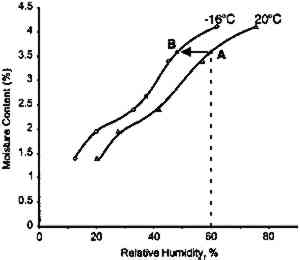
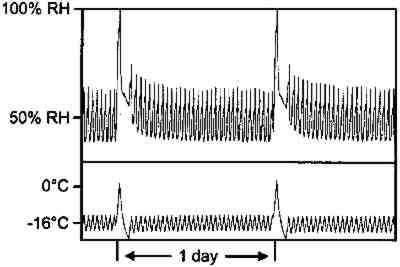
This analysis suggests that the change in some physical properties of gelatin at elevated temperatures is not significantly influenced by the glass transition temperature. This is not to imply that the degradation of these properties is not influenced by Tg, but that the effect is 3.2 IMPLICATIONS FOR LOW-TEMPERATURE STORAGEFor more than 25 years it has been recognized that low-temperature storage will prolong the image life of chromogenic color films (Adelstein et al. 1970). It will also increase the stability of cellulose triacetate base (Adelstein 1992a, 1992b). This has been the repeated and consistent recommendation since that time (Kach 1978; Wilhelm 1978; Eastman Kodak Co. 1983; ANSI 1993). Two types of low-temperature storage environments have been recommended. For relatively small collections, the use of refrigerators or freezers, specifically frost-free units, are advised (Wilhelm 1978). However, for large collections, prolonging the image life has been accomplished using cold storage vaults that operate at moderate to low relative humidities (Goodrich 1980; Wilhelm 1993). Each will be discussed in turn. 3.2.1 FROST-FREE FREEZERSThe recommended procedure for using frost-free freezers is to seal the photographic material in a vapor-tight package at room temperature prior to putting it into a freezer compartment. It has also been recommended that the photographic contents be first moisture conditioned to about 50% RH before being sealed in the package. However, examination of figure 18 indicates that a moisture conditioning procedure is not necessary unless the room temperature humidity is greater than 60% RH. Film in a moisture-tight, sealed package at 20�C and 60% RH (point A in fig. 18) will have an equilibrium relative humidity around 50% RH if the temperature is lowered to −16�C (point B). However, if film is at a room-temperature moisture equilibrium much higher than 60% RH, this treatment could result in the Tg of the gelatin being exceeded during a warm-up period, particularly if the temperature is close to 40�C (table 1). When the gelatin is above Tg, the emulsion layer is more susceptible to physical damage such as ferrotyping or sticking.
It would certainly be less labor-intensive and more user-friendly if photographic film could just be placed in a freezer without vapor-tight packaging. The argument for this approach is that many frost-free cold chambers maintain a moderate relative humidity, with only periodic excursions to a
An additional benefit of enclosures is that they avoid the possibility of moisture condensation on the film during the warm-up period. 3.2.2 REFRIGERATORSRefrigerators do not offer the same cost-benefit advantages as freezers. However, refrigerators above the freezing point (2 to 5�C) can be recommended because of the marked advantages they provide over room temperature storage. Under normal operating conditions, high humidity should not be a concern. However, in commercial equipment with an attached freezer unit there is the possibility of high moisture levels in case of power failure and equipment malfunction. For this reason the photographic material should be enclosed in moisture-protective (but not necessarily moisture-proof) packaging such as cans or polyethylene bags. These will also prevent moisture condensation on the photographic material during the warm-up period. Cardboard boxes are not recommended. 3.2.3 COLD STORAGE VAULTSThe same factors that were considered for freezers also apply to cold storage vaults. However, the latter may be equipped with controls that ensure that the humidity does not increase to unacceptable levels. From the data presented in figure 18, it is recommended that the maximum relative humidity be set at 40% RH. During storage the enclosure needs only to provide physical protection. However, there must be some moisture protection during the warm-up period. An additional polyethylene bag should suffice. |