INVESTIGATION OF A SURFACE TARNISH FOUND ON 19TH-CENTURY DAGUERREOTYPESLEE ANN DAFFNER, DAN KUSHEL, & JOHN M. MESSINGER
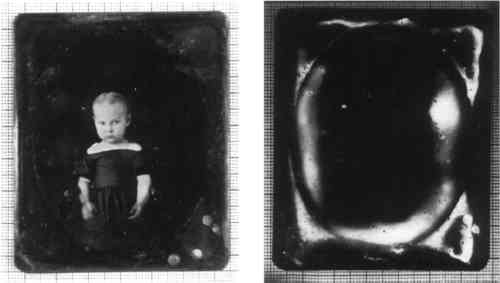
ABSTRACT—A specific tarnishing pattern was first observed in 1992 in a large collection of daguerreotypes viewed under short-wave ultraviolet illumination. While daguerreotypes have been closely inspected under visible light, there appears to be no report of the use of short-wave ultraviolet illumination, a straightforward examination and documentation technique available to conservators. The scope of this investigation includes preliminary analysis, characterization of the tarnish, and a study of its rate of occurrence on a group of 110 daguerreotype plates. Two methods of instrumental surface analysis were used in this study: scanning electron microscopy and Fourier transform infrared analysis. These useful, nondestructive analytical methods are available to the conservator for examination and documentation and provide both inorganic and organic data. The findings indicate that the presence of this fluorescing tarnish may provide evidence of specific past treatments applied to daguerreotype plates. The fluorescing tarnish can serve as a monitoring guide for measuring the deterioration in daguerreotype plates. 1 INTRODUCTIONA vast body of literature exists on 19th-century daguerreotype technology, extending back to the announcement of the daguerreotype in 1839. In the last 15 years, conservators and scientists have studied the complex modes of deterioration of the silver and silver amalgam image of the daguerreotype. One very important aspect of these artifacts is the surface tarnishing of the plate, but due to the extreme thinness of the tarnish layers, this aspect is difficult to examine and analyze. Recent published research by Barger and White (1991) investigated daguerreotype surface corrosion using atomic emission spectroscopy, auger electron spectroscopy, energy dispersive x-ray spectroscopy, Fourier transform infrared spectroscopy, Raman spectroscopy, x-ray diffraction, and scanning electron microscopy. This research followed important earlier research by Swan et al. (1979). The use of ultraviolet illumination in the examination of daguerreotypes was investigated as part of the stabilization and rehousing of the Walter Johnson Collection of postmortem daguerreotypes, a project carried out at the Strong Museum in Rochester, New York, during the summer of 1992. It followed the example of the George Eastman House's condition and monitoring project for its Southworth and Hawes Collection of daguerreotypes. However, unlike the George Eastman House project, both long-wave and short-wave ultraviolet radiation were investigated. This study is an investigation into and preliminary analysis of one surface phenomenon observed during this project on 19th-century daguerreotype plates. The phenomenon is characterized by a bright fluorescence when illuminated by short-wave ultraviolet radiation. It was first observed by the authors in July 1992 at the Art Conservation Department, State University College at Buffalo. It has not been established if the phenomenon is actually a surface tarnish, corrosion product, or accretion, but for now we are referring to it as a surface tarnish, as it most closely resembles other tarnish patterns observed on daguerreotypes. 1.1 ULTRAVIOLET ILLUMINATIONUltraviolet radiation extends from about 10 to 400 nm in the electromagnetic spectrum. The ultraviolet portion of the spectrum is divided into four sections, two of which are used for the examination of artifacts. The most commonly used section is long wave, or UVA, which extends from 320 to 400 nm. The less frequently used section is short wave, or UVC. It extends from about 200 to 280 nanometers in wavelength (Eastman Kodak 1968). Ultraviolet cannot be seen by the human eye. Photographic emulsions, however, are sensitive to much of the ultraviolet spectrum, and by using a filter that absorbs all visible light, it is possible to make a photographic exposure of ultraviolet radiation reflected from a subject. The most common use of ultraviolet illumination in conservation, however, is not the documentation of its reflection or absorption, but rather the observation and photographic documentation of the visible fluorescence produced by some materials when excited by it. This visible fluorescence is caused by the absorption of this higher-energy ultraviolet radiation by the electronic structure of atoms and the release of this absorbed energy through conversion into visible light. Visible fluorescence from ultraviolet excitation at certain wavelengths is a characteristic of certain substances. Long-wave ultraviolet radiation is frequently used in paintings conservation, for example, to help distinguish overpaint from original paint or to ascertain the presence of natural resin varnishes, shellac, and pigments such as zinc white. It has also been applied to the examination of artifacts composed of paper, ceramics, and wood. Objects conservators may be more familiar with the use of short-wave ultraviolet. This illumination is used, for example, to identify inorganic materials such as some minerals and glasses. Until recently, the use of long-wave ultraviolet for the examination of photographs has paralleled the uses in examination of paintings and of works of art on paper: identification of coatings, locating areas of foxing, or identification of hand-colored additions of a photographic image. The International Museum of Photography at the George Eastman House, however, began using long-wave ultraviolet illumination in a condition-monitoring and photodocumentation project for the Southworth and Hawes Collection of daguerreotypes in May 1989. 1.2 THE DAGUERREOTYPEThe daguerreotype was the first commercially popular and successful form of photography. From its announcement in Paris in 1839, the making of daguerreotypes quickly evolved into a precise technology, employed by the scientist, the artist, and layperson. Between 1839 and the late 1850s, thousands of daguerreotypes were produced all over the world. The process was especially popular in America, where 2,000 persons were reported to be practicing the art by 1850 (Taft 1938). The fascinating history of the process has been well documented elsewhere and will not be addressed here (e.g., Taft 1938; Gernsheim and Gernsheim 1968; Buerger 1989; Barger and White 1991). The daguerreotype plate structure consists of a silver layer adhered to a copper substrate. The copper and silver layers were prepared by the cold roll-cladding method (Barger and White 1991). Beginning in the early 1850s, daguerreotype plates were also electroplated, a process that added a layer of silver to the roll-clad silver. This electrolytic silver layer was thought to increase the hardness of the surface, thus enabling the daguerreotypist to achieve a superior polish. The polished plate would be processed by sensitizing with iodide and bromine vapors, exposing the plate in the camera, developing with warm mercury vapors, and, finally, fixing with sodium thiosulfate. The final image of a daguerreotype is composed of silver amalgam particles and silver. The highlight and midtone amalgam particles range in size from .1 to 1 μm in diameter (Swan et al. 1979; Barger and White 1991); the exact structure and size of the particles vary from plate to plate and from point to point on individual plates. The darkest areas of the image remain predominantly unaltered, pure polished silver; thus, if the plate is held so that it reflects back to the viewer a dark background, these areas will appear dark. The amalgam particles forming the midtone and bright areas of the subject scatter light diffusely, and thus these areas will appear lighter in tone. As might be expected, viewing the plate incorrectly so that the background reflects back a bright surface will cause the image to appear as a negative. The most common forms of deterioration found on daguerreotype plates, as cited by Swan et al. (1979), and Barger and White (1991), and also observed by the authors, are: (1) tarnish in the form of silver sulfide from air and pollutants; (2) physical damage such as finger smudges, brush marks, scratches, and gouges; (3) corrosion products from copper erupting from the substrate; and (4) localized accretions of debris from glass and case materials and deposits and residues of polishing compounds. Fine detail and deep, rich shadows in the final image were achieved in part by a superior polish to the silver surface; scratches or blemishes in the surface affect the reflectance, causing physical and visual disruptions to the image. In addition, the silver amalgam particles forming the highlights are extremely delicate and easily abraded; physical damage to these highlights will result in localized loss of the image. Gold toning or “gilding” of many daguerreotypes enhanced the image by strengthening the detail and contrast in addition to improving the permanence of the final image layer. Like the surfaces of other silver objects, daguerreotypes will tarnish if not protected from the environment. The development of complex corrosion products can also develop throughout the daguerreotype structure. Imperfections in the plating and polishing processes can leave tiny pinholes or polishing residues in the surface. Over time, these sites can become the nuclei for copper corrosion or tarnish blemishes (Swan et al. 1979). Historical enclosures, evolved from case housings of 18th-century miniature portraits, were used to protect the delicate silver surface from both physical abrasions and tarnishing. 2 DISCOVERY OF THE FLUORESCING TARNISHDuring the initial stages of the Johnson Collection stabilization project, several techniques for examining and photographing daguerreotypes were investigated at State University College at Buffalo, including the use of both long-wave and short-wave ultraviolet. At this time a brilliant fluorescence was first observed on several unhoused daguerreotype plates viewed under short-wave ultraviolet illumination. It is interesting and important to note that the areas that fluoresce are not always discernible when the plate is examined under visible light. 3 DESCRIPTION OF THE FLUORESCING TARNISHA long-wave ultraviolet lamp can be used to accentuate surface anomalies on daguerreotypes, such as scratches and applied color, as observed at the Eastman House. The reflection of visible blue light that leaks through the long-wave lamp's filter seems to help highlight subtle variations in the surface. Long-wave illumination does not reveal more information about the plate's condition with respect to observed fluorescence, however, because the daguerreotype plates do not emit visible fluorescence. On the other hand, using short-wave ultraviolet illumination, a spectacular fluorescence can be seen on some daguerreotype plates (fig. 1).
Because glass absorbs short-wave ultraviolet radiation, bound, encased daguerreotypes cannot be examined with short-wave ultraviolet illumination. A rehousing and photo-documentation project such as the one at the Strong Museum thus provides an ideal opportunity to survey and record this phenomenon. The 79 daguerreotypes examined at the Strong Museum are part of the Walter Johnson Collection of postmortem and mourning-related objects. The Johnson daguerreotypes are the single largest known collection of postmortem daguerreotype portraits in the United States. The collection, assembled over a 20-year period by Johnson, focuses on the postmortem portrait most commonly encountered in the 19th century. It also includes 19th- and 20th-century examples of memorial cards, lithographic prints, mourning jewelry, advertisements, and literature from Johnson's reference library. Only 15 of the daguerreotypes surveyed had older or original seals. Twenty-one were sealed with modern tapes, including masking tape and clear, pressure-sensitive tapes. The remaining daguerreotypes were not sealed at all, although evidence of previous tapes was observed on the verso of these plates. Seventy plates in the Johnson Collection showed signs of some previous cleaning attempts. Overall the collection was in fair to good condition, considering the degree of handling, unbinding, and rebinding of the plates and cleaning that had been undertaken (Daffner 1992). Twenty-one daguerreotypes in private collections and 10 French daguerreotypes from the George Eastman House were also examined, bringing the total to 110 plates. Of these 110, 50% exhibited the fluorescing tarnish. The fluorescence appears to occur primarily in three forms, although many variations were observed. 3.1 EDGE TARNISHThe fluorescence is most commonly present at the very edge of the plate, although under normal illumination it is not always possible to discern the exact location of the fluorescing region. The presence of the fluorescence at the edge of the plates may correspond to those areas that were not completely toned or gilded during the final processing procedures (fig. 2).
At the height of the daguerreotype's popularity, the process of gilding the daguerreotype image was a regular procedure. The most common technique used by operators was to hold the plate, image side up, parallel to the table, over an alcohol lamp. A solution of gold chloride was poured onto the image side of the plate and contained there by surface tension. When the gilding was complete, excess solution could be poured off at one corner. Except at the point of pouring off, the very edges were often not completely covered with the gold chloride solution. In our study, fluorescence was observed to be associated with these nongilded edges, which were often tarnished. 3.2 RINGSThe fluorescence also appears as rings. There can be a single ring or several concentric rings present at a single location (figs. 3, 4). These rings are sometimes visible under normal illumination as a very light orange stain. Small accretions at the nucleus of rings were occasionally observed.
3.3 CONTINUOUS FILMSIn several examples, the fluorescence was also observed as a continuous film. In these instances, it was not always possible to see the location of the fluorescence under normal light (see fig. 1). 3.4 OPENING OF METAL MATThe fluorescence was observed in some plates to be present only within the opening of the decorative metal mat (fig. 5). The cover glass, however, does not appear to have a direct effect on the development of the fluorescence, as on other plates the tarnish was found underneath metallic mats where there was no contact between the cover glass and the daguerreotype plate.
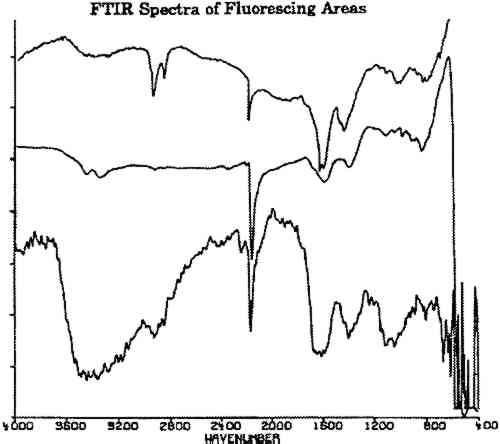
The following additional observations were made: On some plates, the fluorescence is related to pits, scratches, or losses in the surface silver. Fluorescence was seen to be associated with the addition of gold flake coloring, primarily occurring directly adjacent to the area of application. Ninety American daguerreotypes were examined, of which 47 showed fluorescence. Ten French daguerreotypes from the George Eastman House were also examined. The fluorescence was observed on three of these plates. On two plates examined, the fluorescence was observed alongside unidentified fibrous accretions. The variety of patterns and relative degrees of fluorescence observed on the plates suggest a number of causes for the fluorescence. 4 DOCUMENTATIONBefore beginning the instrumental analysis, selected daguerreotypes were photographed with 4 � 5 black-and-white Kodak Plus-X film at 1x magnification in normal light and under short-wave illumination using a Raytech Model SW-218 UVC illuminator. A no. 8 Wratten gelatin filter was used to filter out reflected ultraviolet radiation and blue light. These photographs were made to aid in documenting the location of the fluorescing material during our analysis and to serve as a permanent record for future monitoring. In addition, to record the fluorescence color, the plates were photographed with 35 mm Kodak Ektachrome daylight 200 speed reversal film (ED-135) using the same short-wave ultraviolet unit. Kodak 2E and CC40R Wratten gelatin filters were used as standard filtration of reflected ultraviolet radiation and blue light. 5 SOLVENT TESTSA series of basic solvent tests were conducted on one of the plates exhibiting fluorescence. The spot tests were conducted in a fume hood under UVC illumination. The fluorescence was immediately eliminated by xylene and N,N-dimethyl formamide, and remained so after complete evaporation of the solvent. The fluorescence was not affected by water, ethanol, 1 M HCL, or ethylene diamine tetraacetic acid (EDTA) trisodium salt. 6 SCANNING ELECTRON MICROSCOPEDue to the delicate nature of the daguerreotype surface and the extremely thin tiers of tarnish and oxidation, it was necessary to use methods of analysis that are both nondestructive and suitable for thin-layer analysis. The first choice was scanning electron microscopy (SEM). The surface topography of daguerreotype plates has been well documented with SEM (Swan et al. 1979; Barger and White 1991), and this method is available to many conservators. Small plates fit inside the vacuum chamber of the scanning electron microscope, with no harm to the artifact. Daguerreotype plates (figs. 3, 4) were examined with a Hitachi Instruments, Inc. S-800 field emission scanning electron microscope equipped with a Princeton Gamma Tech IMIX microanalysis system for energy dispersive x-ray analysis. The following x-ray spectra results were obtained in one area unassociated with the fluorescence and two areas associated with fluorescence: (1) Spectra from areas of the plates that did not fluoresce under UVC irradiation showed one substantial silver peak. Smaller peaks of gold and mercury were also present. (2) Spectra obtained from an accretion present at the center of a fluorescent ring showed one substantial peak of copper, with smaller peaks of silver, chlorine, and iron. (3) Spectra from the fluorescing ring showed one substantial peak of silver, accompanied by smaller peaks of copper. Trace peaks of iron and mercury were also indicated. These results suggested that the unknown fluorescing material is primarily composed of silver and copper. With the exception of the trace iron elements, these inorganic elements comply with the typical daguerreotype plate structure. Swan et al. (1979) singled out similar spots during their examination of daguerreotypes using SEM. They described “thin orange and iridescent films” and rings of orange films containing copper and traces of iron, surrounded by a clear ring, then a second wider ring of orange tarnish that also contains copper. The rings and films Swan et al. analyzed using SEM have spectra similar to the fluorescing areas investigated in this study. 7 FOURIER TRANSFORM INFRARED SPECTROSCOPYFourier transform infrared spectroscopy (FTIR) yields data on organic and inorganic compounds present on a sample. For analysis, a bare daguerreotype plate was simply placed on the platform of the infrared microscope. No contact was made between the microscope and the plate, avoiding damage to the surface. The three spectra shown in figure 6 were obtained on the fluorescing areas of two daguerreotypes (figs. 2, 4) using a Nicolet 710 spectrometer equipped with an IR-PLAN infrared microscope. The uppermost spectrum in figure 6 was taken from a fluorescing edge. The two lower spectra in figure 6 were taken from two concentric rings on the daguerreotype in figure 4, one of the daguerreotype plates that was used for the SEM analysis. The spectrometer was operated in the reflectance mode using a 15� cassegrainian infrared objective, at 8 cm−1 resolution.
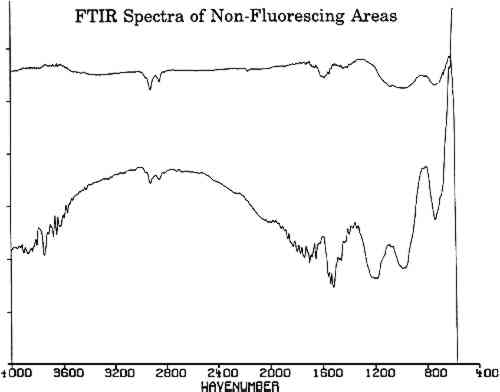
All three spectra of figure 6 show a strong absorption band at 2165 cm−1, which is not present in spectra of adjacent, nonfluorescing areas (fig. 7). This absorption band is within the wave number range in which the cyanide ion and the carbon-nitrogen triple bond bands groups absorb. Since there are no other common organic groups showing a strong absorption in this region other than a carbon-nitrogen triple bond, one plausible explanation is that the observed absorption is a cyanide compound. The spectra of the fluorescent deposits indicate that the residues are mixtures, composed of what appears to be a cyanide compound and the salt of an organic acid. The infrared analysis cannot identify the specific cation associated with the cyanide ion. The cyanide absorbances, however, are consistent with the absorbances expected for either potassium or gold cyanide (Bellamy 1968). Elemental analysis techniques such as micro x-ray diffraction might be able to identify the material if it is crystalline. If the crystals are too fine, however, this technique will not provide an adequate diffraction pattern for positive identification.
8 TENTATIVE IDENTIFICATION OF THE FLUORESCENT MATERIALThe detection of copper and cyanide ion in the SEM and FTIR studies of the fluorescent areas suggests that the fluorescing material might be a copper cyanide compound. This possibility is supported by the observation that, after being separately immersed in aqueous 0.2 M sodium cyanide for .5 minute and then removed, a copper plate fluoresced under UVC irradiation whereas a silver plate did not. In addition, spot tests on several areas of the fluorescing tarnish on actual plates indicated the fluorescing material to be insoluble in water. Although this result is consistent with a water insoluble copper cyanide compound being the fluorescent material of the spots, possibly cuprous cyanide, further analytical work will be necessary to establish this point unambiguously. 9 CYANIDE IN THE DAGUERREOTYPE PROCESSThe historical literature mentions six procedures in which cyanide was used in the daguerreotype process: electroplating; gilding or gold toning; brightening; fixing; “engraving by Galvanism”; and cleaning. The following is a brief outline of these processes. 9.1 ELECTROPLATINGElectroplating, galvanizing, or resilvering a daguerreotype plate was a common procedure used to improve the surface properties of the daguerreotype plate by allowing it to be more finely polished. The result was greater detail and deeper shadows in the final image. By 1853 it was a regular practice to plate silver by electrolytic action using a Smee's or Daniell's battery (Barger and White 1991). In this process, an unprepared copper or roll-clad plate is treated by adding a uniform layer of silver on the surface of the plate by electrolytic action. Electroplating was the first procedure in which cyanide may have been introduced in the preparation of the daguerreotype plate: silver was electroplated in the cell in a solution of potassium cyanide. Daguerreotypists were also known to recycle used plates by resilvering using this method. 9.2 GILDING OR GOLD TONINGGilding or gold toning procedures were thought to improve image stability, image contrast, and resistance to corrosion. One gilding procedure referred to in the 19th-century literature was known as “tinging daguerreotypes” (Napier 1864). A patent for gilding daguerreotypes was taken out on October 26, 1852, by Charles L'Homdieu, a daguerreotypist from Charleston, South Carolina. According to Rinhart and Rinhart (1981), this process employed a solution of hot gold cyanide. Barger and White (1991) indicate the solution was composed of sodium thiosulfate and gold chloride. 9.3 BRIGHTENINGThe daguerreotypist sometimes immersed the plates in a solution of potassium carbonate and potassium cyanide with “a little alum, borax and some other things” (Rinhart and Rinhart 1981, 188). This procedure may have brightened the highlights by slightly etching them. 9.4 FIXINGBarger and White (1991) note that cyanide solutions were sometimes used in the actual fixing procedures. 9.5 ENGRAVING BY GALVANISMThis was a rare daguerreotype procedure, although oddly it was frequently referred to in the 19th-century electro-metallurgy literature. This procedure created electroplate copies of daguerreotypes using an electrolytic cell and a solution of gold cyanide and potassium cyanide (Napier 1864, 493). 9.6 CLEANINGPotassium or sodium cyanide solutions were used to remove tarnish from daguerreotype plates throughout the 19th century and until the 1950s, when thiourea was introduced as a less toxic alternative for removing silver sulfide tarnish from daguerreotypes (Newhall 1961). 9.7 THE USE OF CYANIDEThese are six examples of how cyanide might have been employed by the daguerreotypist, and while other uses may exist, this list illustrates the frequency and ease whereby cyanide could have been used at any stage in the production of daguerreotypes. It is important to keep in mind that the methods of each daguerreotypist could differ greatly from the published literature; basic procedures were adapted to fit individual situations, and variations of these techniques were often employed. 10 SUMMARY OF OBSERVATIONS AND FINDINGSOne hundred and ten daguerreotypes have been examined in this study under short-wave ultraviolet illumination. Of these, 50 showed some degree of visible fluorescence. The plates that showed signs of overcleaning, prolonged exposure to the environment, and general mishandling had developed advanced stages of the fluorescence. Several plates that retained old seals virtually intact also showed fluorescence, but only to a very slight degree. While no definitive conclusion can be made at this time, a direct correlation may exist between the extent of handling and exposure to the environment with the degree of fluorescence present. The following list summarizes the principal observations of this study:
11 CONCLUSIONSShort-wave ultraviolet examination yields a uniquely discernible fluorescence pattern on some daguerreotype plates. The origin of this fluorescence has not been established conclusively, nor has the stability of the fluorescing material. Nevertheless, short-wave ultraviolet documentation can be part of active monitoring programs for daguerreotypes. Documentation of this fluorescence may prove to be an extremely useful method for indicating the progression of tarnishing patterns. The carbon-nitrogen triple bond bands present in the FTIR spectra are associated with different salts. Identification of the various fluorescence patterns may provide evidence of the technical history of individual plates. As described above, there are at least six ways in which cyanide could have been introduced into the daguerreotype process. The procedure of examining daguerreotypes under short-wave ultraviolet radiation requires special safety considerations during handling for both the object and the examiner. When unbound, the delicate daguerreotype plate is extremely vulnerable to surface abrasion. The photographing of bare, unprotected plates should be conducted only by an experienced photographer, preferably in the presence of a conservator. During examination, extreme caution should be used when handling bare plates that have shown fluorescence due to the highly toxic nature of cuprous cyanide and silver cyanide. While areas of copper cyanide compounds may be identified by fluorescence, silver cyanide is a grayish white to black powder that could easily go undetected on the multifaceted terrain of the daguerreotype surface. Safety precautions should be undertaken, such as wearing ultraviolet-absorbing safety glasses to protect the eyes and latex examination gloves to prevent contact with cyanide. Inhalation of surface debris should be avoided at all costs, as the cyanide compounds are fast acting, deadly poisons (Strahan 1980). It was noted that the plates examined that had remained adequately sealed did not show significant development of the tarnish. Therefore opening the plate package is not recommended unless the seals are inadequate and need to be replaced or a thorough, systematic monitoring project is to be undertaken for the entire collection. The complex nature of the daguerreotype has yet to be fully understood. Little by little, we are gaining insight to its structure and deterioration to aid in its preservation and appreciation. Interpretation of the fluorescence observed on daguerreotype plates deserves further attention and research, and we hope this investigation will inspire other conservators and scientists to explore this phenomenon. ACKNOWLEDGEMENTSThe SEM studies were carried out by Peter Bush, director, Biomaterial Instrument Center, State University College at Buffalo. The FTIR spectra were obtained by Sharon Markel of the Eastman Kodak Analytical Technology Department, Eastman Kodak Company, Rochester. We are grateful to both for having given many hours of their time to this project. The authors would like to thank the Strong Museum and conservation staff at the George Eastman House for the use of their facilities. We are grateful to Grant Romer, director of museum practices and conservation at the George Eastman House, for first suggesting the use of long-wave ultraviolet radiation for examining daguerreotypes and for his assistance in this study. We would also like to thank F. Christopher Tahk, director of the Art Conservation Department of the State University College at Buffalo, for expert guidance and the use of his daguerreotype collection. REFERENCES
Barger, S. M. and W. B.White. 1991. The daguerreotype nineteenth-century technology and modern science. Washington, D.C.: Smithsonian Institution Press. Bellamy, L. J.1968. Advances in infrared group frequencies. London: Methuen. Buerger, J. E.1989. French daguerreotypes. Chicago: University of Chicago Press. Daffner, L. A.1992. Swinney internship report. Strong Museum, Rochester, N.Y. EastmanKodak. 1968. Ultraviolet and fluorescence photography. Eastman Kodak Publication M-27. Rochester: Eastman Kodak. Gernsheim, H., and A.Gernsheim. 1968. L.J.M. Daguerre. New York: Dover Publications. Napier, J.1864. A manual of electro-metallurgy: The application of the art to manufacturing processes. Philadelphia: Henry Carey Baird. Newhall, B.1961. The daguerreotype in America. New York: Dover Publications. Rinhart, F., and M.Rinhart1981. The American daguerreotype. Athens, Ga.: University of Georgia Press. Strahan, D.1980. Treatment of a silver dragon for the removal of silver cyanide and chalconatronite. Journal of the American Institute for Conservation25:73–80. Swan, A., C. E.Fiori, and K.F.J.Heinrich. 1979. Daguerreotypes: A study of the plates and the process. Scanning Electron Microscopy1:411–23. Taft, R.1938. Photography and the American scene. New York: Macmillan Company. FURTHER READINGde la Rie, R.1982. Fluorescence of paint and varnish layers (parts 1 and 2). Studies in Conservation27:1–7, 65–69. Eder, J. M.1978. The history of photography. New York: Dover Publications. Griffiths, P. R.1983. Fourier transform infrared spectrometry. Science222:297–302. Pobberovsky, I.1971. Study of iodized daguerreotype plates. Rochester: Graphic Arts Research Center. Swan, A.1981. The preservation of daguerreotypes. Postprints, American Institute for Conservation 9th Annual Meeting, Philadelphia. Washington, D.C.: AIC. 164–72. Walls, J. M.1989. Methods of surface analysis. Cambridge: Cambridge University Press. AUTHOR INFORMATIONLEE ANN DAFFNER holds a B.A. in fine art printmaking from San Francisco State University and an M.A. and certificate of advanced study in conservation (1994) from the State University College at Buffalo. She is currently an Andrew W. Mellon Fellow in conservation at the Metropolitan Museum of Art in New York. Address: Paper Conservation Department, Metropolitan Museum of Art, 1000 Fifth Ave., New York, N.Y. 10028. DAN KUSHEL received an M.A. in 1972 and completed doctoral course work in art history at Columbia University. He also holds an M.A. and certificate of advanced study in conservation (1976) from the State University College at Oneonta (Cooperstown Graduate Program in Conservation). He served as assistant conservator at the Brooklyn Museum and in 1978 joined the faculty of the Cooperstown Graduate Program (now the Art Conservation Department, State University College at Buffalo), where he currently holds the rank of professor, teaching in the areas of technical examination and documentation and paintings conservation. Address: Art Conservation Department, Rockwell Hall 230, State University College at Buffalo, 1300 Elmwood Ave., Buffalo, N.Y. 14222. JOHN M. MESSINGER II received his Ph.D. in organic synthesis from the State University College at Buffalo in 1986. That year, he joined the faculty of the Art Conservation Department of the State University College at Buffalo as assistant, and then associate, professor of conservation science. In 1993, he resigned his position at the college to enter the School of Dental Medicine of his alma mater to pursue studies leading to the DDS. Address: As for Kushel.
 Section Index Section Index |