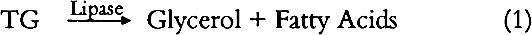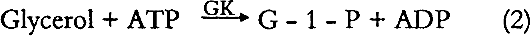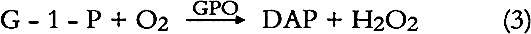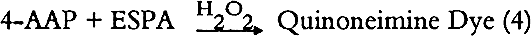BINDING MEDIA IDENTIFICATION IN PAINTED ETHNOGRAPHIC OBJECTSDUSAN STULIK, & HENRY FLORSHEIM
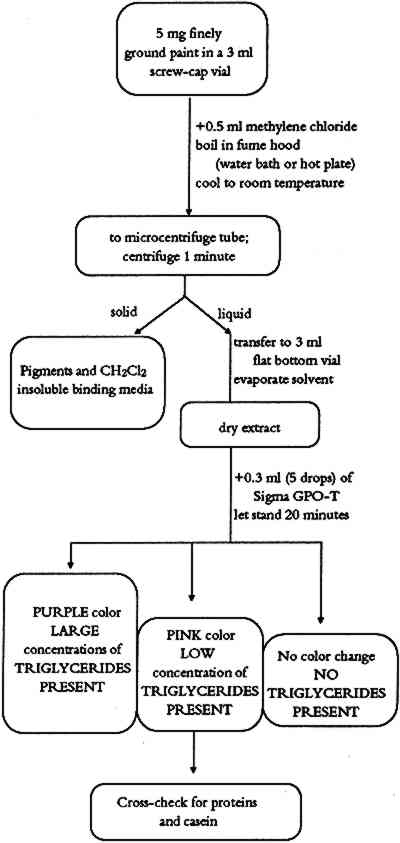
ABSTRACT—This article describes a binding media identification procedure for the analysis of 5 mg samples from painted ethnographic objects. It is based on modified medical diagnostic and forensic science tests and is designed to identify vegetable oils, lards, simple sugars, tree gums, starches, animal glues, casein, eggs, and blood in paint layer samples. Flow diagrams for individual tests are described as well as a binding media identification procedure and a general binding media identification flow chart. 1 INTRODUCTIONIdentification of binding media in the painted layers on ethnographic objects is an important step prior to any conservation treatment. The first attempts to paint with colored materials were made without using any binding media or using only a temporary binding medium (water). Eventually craftsmen and artists realized that such procedures did not give satisfying or long-lasting results. They recognized the need for binding media that would hold pigment particles together and adhere the pigmented layer to the substrate. The problem of identifying binding media in ethnographic objects is complex. Each culture and each time period had a set of favorite media. The range of binding media used on ethnographic objects is broad and covers a variety of materials from tree gums, salmon eggs and orchid juices to modern synthetic polymers. Binding media found on ethnographic objects are raw natural products (egg, honey, or blood), processed natural products (animal glue, casein, starch, or oil), or synthetic organic materials. This paper focuses on natural binding media that chemically belong to groups of lipids (vegetable oils, or lard), carbohydrates (honey, tree gums, or starches), and proteinaceous materials (animal glue) and on complex binding media (egg, blood, or fruit juices). For example, egg is a complex binding medium that contains proteins, oils, and carbohydrates. Blood contains proteins, oils, carbohydrates, and hemoglobin. Several simple chemical tests have been modified for identification of binding media in paint layers of art objects. These tests are based on qualitative spot tests (Feigl 1966), microchemical analytical procedures (Schramm and Hering 1988), or chemical microscopy methods (Masschelein-Kleiner 1986). The literature mentioned serves as a good introduction to these methods and contains references needed for more detailed study. The aim of this study was to develop a simple binding media identification system that would not require extensive training to master and would not use sophisticated chemical and physical instrumentation, which is beyond the means of many conservation laboratories. Because of their simplicity and widespread availability, commercial test kits developed for modern medical diagnostics are the basis for much of our work. Some additional tests were added using methods of qualitative analytical chemistry and forensic science. We have made every effort to include test reagents that are readily available worldwide. Some tests included in the binding Each test was checked for possible interference from the other binders considered in this study. The possible effect of some pigments important in ethnographic art objects was studied; they include yellow and red ochre, kaolin, limestone, gypsum, ultramarine, charcoal, manganese dioxide, and “battery black” (an impure form of manganese dioxide extracted from old batteries by Australian and African ethnographic artists and used after World War II). Where interference was observed, it is noted under the comments for the individual tests. 2 TEST FOR TRIGLYCERIDESTriglycerides (TG) used as binding media on ethnographic art objects can be categorized as oils or lards. Chemically, both oils and lards belong to a class of chemical compounds known as esters. Esters are produced when an alcohol and a carboxylic acid undergo a condensation reaction. Vegetable oils are obtained from seeds and nuts of certain plants. Lard is a solid fat prepared by rendering fatty tissue of animals. Both vegetable oils and lards are composed of triglycerides of fatty acids (esters of glycerol and higher saturated or unsaturated carboxylic acids). The number of carbon-carbon double bonds in the triglyceride affect the drying properties of the material. Drying oils contain a large concentration of polyunsaturated fatty acids (especially linolenic acid). Nondrying oils contain only very small amounts of this acid and consist of varying amounts of saturated and mono- or disaturated fatty acids. Lards are composed of palmitin, stearin, and olein (triglycerides of palmitic, stearic, and oleic acid, respectively). Hydrolysis of oils and lards produces glycerol (glycerine) and free fatty acids. The fact that glycerol is an integral part of all vegetable oils and animal lards serves as a basis for a laboratory proof of these materials in paint layers. 2.1 METHODThe sample is dissolved in hot methylene chloride. Insoluble material is removed by centrifugation. The methylene chloride solvent is evaporated, and the presence of glycerol in the sample is determined by an enzymatic test. Triglycerides are first hydrolyzed by lipoprotein lipase to glycerol and free fatty acids.
Glycerol is then phosphorylated by adenosine triphosphate [ATP] forming glycerol-I-phosphate [G - 1 - P] and adenosine-5-diphosphate [ADP] in the reaction catalyzed by glycerol kinase [GK].
G - 1 - P is then oxidized by glycerol phosphate oxidase [GPO] to dihydroxyacetone phosphate [DAP] and hydrogen peroxide [H2O2].
Hydrogen peroxide catalyzes the reaction of 4-aminoantipyrine [4-AAP] with sodium N-ethyl-N-[3-sulfopropyl]-m-anisidine [ESPA] to form a quinoneimine dye.
A pink to purple color of the quinoneimine dye indicates the presence of triglycerides in the analyzed sample (McGowan et al. 1983; Barham and Trinder 1972). 2.2 PROCEDUREThe flow chart for the triglyceride test is shown in figure 1. A finely ground paint
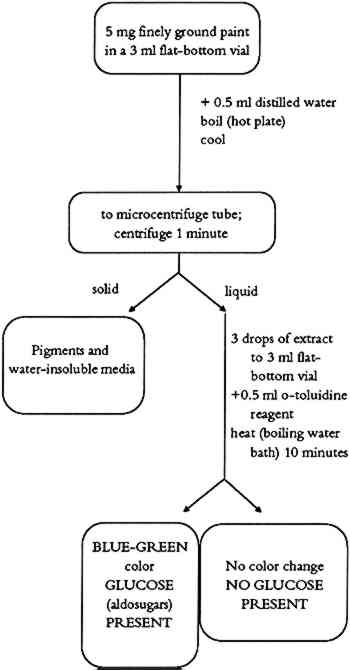
TABLE 2 BINDING MEDIA TEST REAGENTS 2.3 COMMENTSPaint samples containing drying oils, animal fat (lard), egg yolk, or unpurified milk casein give a positive color. The color intensity of the test varies with concentration of triglycerides in the sample. Egg yolk and whole milk (source of casein) contain some triglycerides and may give a very weak positive test. If weak tests are obtained, the sample should be tested for presence of protein and phosphorus. Paint samples treated with glycerine or with material containing glycerine might also give a positive test for triglycerides even without any triglycerides present. 3 TESTS FOR CARBOHYDRATESComplex carbohydrates, (polysaccharides such as gums) and simple sugars (monosaccharides such as honey) have been used as binding media in ethnographic objects. In the past, the term “gums” was used as a general term to denote a Honey is the sweet, sticky material made from flower nectar by certain bees. Honey absorbs and retains moisture, thus retarding drying. Honey was used as a plasticizer to improve elasticity of other binding media (glues, or gums) or as a straight binding medium in certain ethnographic objects. All types of honey are rich in the simple sugars dextrose (D-glucose) and levulose (D-fructose). Detection of carbohydrates in paint samples is based on a sugar-specific chemical reaction. 3.1 TEST FOR SIMPLE SUGARS3.1.1 MethodSimple sugars are extracted from the sample using hot water. Upon heating, o-toluidine reacts with aldosugars (glucose) in the presence of acid to form a blue-green colored complex. 3.1.2 ProcedureThe simple sugar test flow chart is shown in figure 2. About 5 mg of finely ground paint sample or residue from the methylene chloride extraction (triglyceride test) is mixed with 0.5 ml of distilled water in a 3 ml flat-bottom vial. The resulting suspension is boiled for a short time on a hot plate at a low heat setting. After cooling to room temperature, the suspension is transferred to a microcentrifuge tube. After about 1 minute of centrifugation, 3 drops of supernatant are mixed in a 3 ml flat-bottom vial with 0.5 ml of o-toluidine reagent. The vial is placed in a boiling water bath for about 10 minutes. A positive test for simple sugars is indicated by the formation of a blue-green color (Hultman 1959; Hyvarinen and Nikkila 1962).
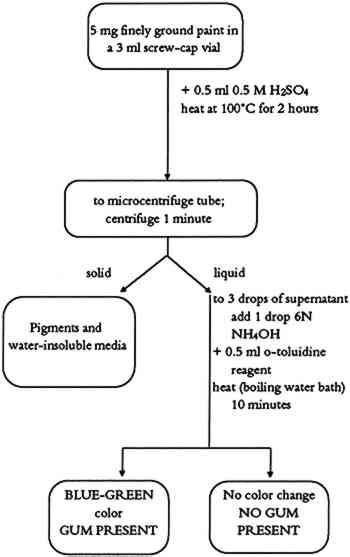
3.1.3 CommentsThis procedure tests for simple sugars (honey) as either a binding medium or a binding medium additive in paint samples. In the case of a very weak positive test, it is important to search for other binding media. Some complex binding media (milk, blood, egg, or plant juices) might contain enough carbohydrates to produce a weak positive test. In such cases, a combination of simple tests or the use of instrumental methods may be necessary. Some pigments (charcoal) were found to interfere with the test. Hence, the sample for the simple carbohydrate test should be obtained from nonblack paint layers of the object to avoid such a problem. 3.2 COMPLEX CARBOHYDRATE (GUM) TEST3.2.1 MethodThis method is identical to the test for simple carbohydrates with the addition of acid hydrolysis of the polysaccharide chains. 3.2.2 ProcedureThe complex carbohydrate test flow chart is shown in figure 3. About 5 mg of finely ground paint sample is mixed with 0.5 ml of 0.5 M H2SO4 in a 3 ml screw-cap vial. The vial is closed tightly and placed in an electric oven preheated to 100�C for 2 hours. The vial is removed from the oven, and allowed to cool to room temperature. The contents of the vial are transferred to a microcentrifuge tube and centrifuged 1 minute. Three drops of the supernatant are placed in a 3 ml flat-bottom vial. One drop of 6N NH4OH is added, followed by 0.5 ml of o-toluidine reagent. The vial is placed in a boiling water bath for about 10 minutes. A positive test is indicated by the formation of a blue-green color.
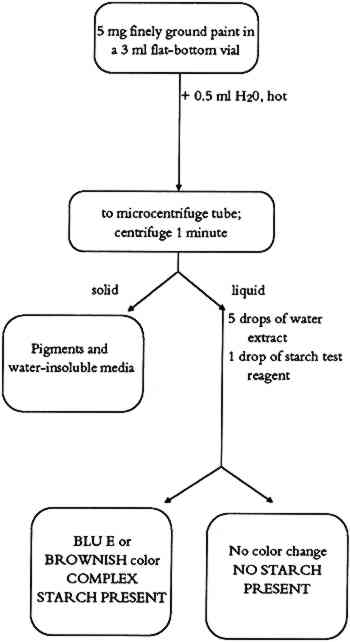
3.2.3 CommentsThis test identifies complex carbohydrates (natural plant gums) in paint samples. Battery black (impure manganese dioxide), yellow, and red ochres were found to interfere, giving a negative test. Samples containing different pigments (preferably white pigments) should be obtained for the complex carbohydrate test. 4 TEST FOR STARCHESStarch is present in all plant cells. Primary sources for starches are potatoes, rice, corn, wheat, and arrowroot. The starch granules are separated from these plants by milling and washing in water. Starch granules contain two polymers, amylase and amylopectin. Chemically, the amylase is a large linear polymer of 1,4-anhydroglucose. Amylopectin molecules are also based on the same anhydroglucose polymer, but they have a large number of short linear chains attached to the main polymer chain. The heating of dry starch to 160�-190�C produces dextrin. Both starches and dextrins are used as paste adhesives. In its more soluble form, starch can also be used as a binding medium for pigments. 4.1 METHODAn aqueous solution of starch gives a blue or brown color in a dilute solution of iodine. The active fraction of starch is amylose. The amylose exists as a coiled helix of poly α -D-glucose into which linear molecules can fit. In the presence of starch, iodine ions (I−) form a long chain of I−5 ions that occupy the center of the amylose helix. It is this dark blue I−5-amylose complex that provides proof of the starch in the paint sample (Vogel 1989). 4.2 PROCEDUREThe starch test flow chart is shown in figure 4. About 5 mg of finely ground paint sample or residue from the methylene chloride extraction is mixed with 0.5 ml of distilled water in a 3 ml flat-bottom vial. The resulting suspension is heated to a low boil on a hot plate. It is boiled for about 2 minutes and then allowed to cool to room temperature. The suspension is transferred into a microcentrifuge tube and centrifuged 1 minute. Five drops of supernatant are mixed with 1 drop of starch test reagent. The presence of starch is indicated by an immediate color change to blue or brown.
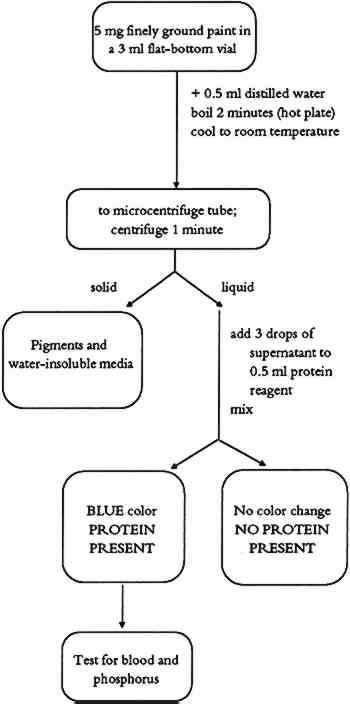
4.3 COMMENTSThis test identifies starch as a binding medium or adhesive in paint samples. The resulting color complex is usually blue, but different shades of brown color also indicate the 5 TEST FOR PROTEINSProteins are simple or complex polymers consisting of chains of amino acids. There are 22 naturally occurring amino acids that are bound together in proteins by peptide linkages (-CO-NH-). The molecular weight of proteins ranges from 104 to 107. Amino acids occur in varying proportions in different proteins. Several proteinaceous materials have been used as binding media in ethnographic art objects. Glue is obtained by drying solutions made from boiling animal skin or bones in water. Gelatin is a purer form of glue made from selected, cleaned animal matter. Egg has two protein-rich parts. Egg white is composed of different proteins (65% ovalbumin). Egg yolk is an emulsion containing water, lipids, proteins (15% livetine and phosvitine), and the phospholipid lecithin. The yellow color is due to the presence of carotene pigments. Casein is the main protein in milk. It is a complex mixture of proteins and phosphoproteins. 5.1 METHODThe protein test is based on the formation of a color complex of proteins with an organic dye (Bradford 1976). In this test, a blue dye complex is formed between Coomassie blue (brilliant blue G) and small amounts of protein in an acid-alcoholic medium. 5.2 PROCEDUREThe protein test flow chart is shown in figure 5. About 0.5 ml of distilled water is added to 5 mg of finely ground paint sample or the residue from the methylene chloride extraction (triglyceride test) in a 3 ml flat-bottom vial. The suspension is boiled on a hot plate for 2 minutes. After cooling to room temperature, the suspension is transferred to a microcentrifuge tube and centrifuged 1 minute. Three drops of the supernatant are added to 0.5 ml of protein reagent in a 3 ml test tube and
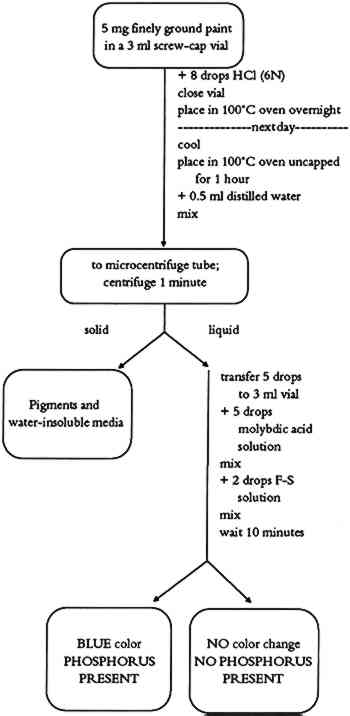
5.3 COMMENTSProtein-containing binding media (animal glue, egg, casein, or blood) are determined with this test. Yellow ochre pigment was found to cause a weakened or negative protein test. Binding media of low protein content (egg yolk, or blood) may give weak positive test results. 6 TEST FOR PHOSPHORUSCertain binding media contain phosphoproteins. In such cases, the detection of phosphorus helps to identify the binding medium. Phosphoproteins are proteins with an ester bond between the side chain of a serine (one of the amino acids found in proteins) and a phosphate unit. Casein, milk, and egg yolk contain enough phosphoproteins that the phosphorus test can be used as a secondary test for binding media identification. 6.1 METHODPhosphoproteins are hydrolyzed using a strong acid. The hydrolysate is then reacted with ammonium molybdate in an acid solution to form phosphomolybdate. A mixture of sodium bisulfite, sodium sulfite, and 1-amino-2-naphtol-4-sulfonic acid (Fiske-Subbarow reducer) is used to reduce the phosphomolybdate to form a phosphomolybdenum blue complex. 6.2 PROCEDUREThe phosphorus test flow chart is shown in figure 6. About 8 drops of 6N HCl are added to about 5 mg of finely ground paint in a 3 ml screw-cap vial. The vial is closed tightly and placed into a 100�C preheated oven overnight. The vial is removed from the oven and cooled to room temperature. The vial's cap is removed, and the vial is placed again into a 100�C oven to evaporate the acid. This takes about 1 hour. About 0.5 ml of distilled water is
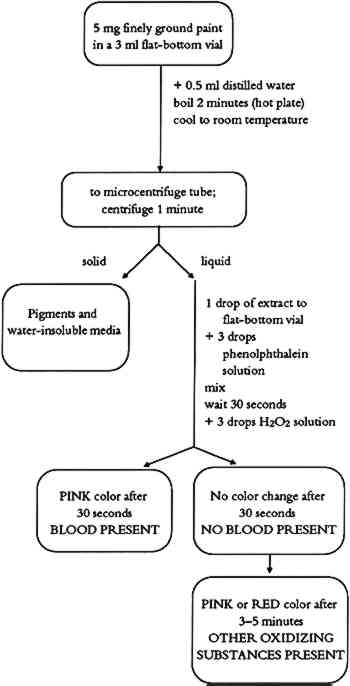
6.3 COMMENTSThe test allows for identification of phosphorus in binding media. Casein gives a strong phosphorous test. If milk or egg yolk was used as a binding medium, the test is weaker due to their lower phosphorus content. 7 TEST FOR BLOODSeveral cultures have been known to use blood as a binding medium for painting. Although blood appears to be a simple liquid, it is actually a body tissue. The cells, instead of being joined together as in solid body tissues, are suspended in a fluid, called plasma. The cellular portion of blood consists chiefly of red blood cells, with a smaller number of white blood cells and platelets. Chemically, blood is made up of water, inorganic salts, proteins, lipids, polysaccharides, glucose, cholesterol, and hemoglobin. This blood test is based on detection of heme molecules in the paint sample and is used in forensic science to detect fresh or aged blood. 7.1 METHODThe test is based on the peroxide-mediated oxidation of reduced phenolphthalein in which the heme molecule acts as a catalyst (Saferstein 1982). 7.2 PROCEDUREThe blood test flow chart is shown in figure 7. About 0.5 ml of distilled water is added to 5 mg of finely ground paint sample in a 3 ml flat-bottom vial. The resulting suspension is boiled for about 2 minutes on a hot plate. After
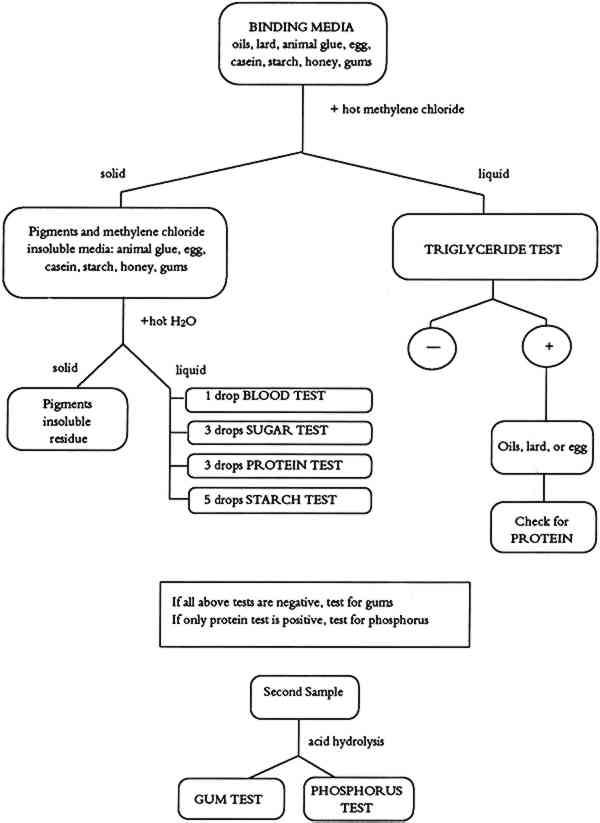
7.3 COMMENTSThe blood test can be used to detect the presence of blood in red or black paint layers. Iron oxides and metal salts have been reported to give a false positive test (Saferstein 1982). Our tests with red ochre did not show such a problem. The color obtained when charcoal black-blood paint layers are analyzed gives a weaker positive test, possibly due to the heme adsorption on charcoal particles. Pink or red color that develops after 3-5 minutes is due to the presence of other oxidizing substances or to peroxidase catalysts from certain plant sources (Saferstein 1982). 8 BINDING MEDIA IDENTIFICATION PROCEDUREIf there is previous knowledge of binding media used by a particular culture in a corresponding time period or in a given geographic region, the Binding Media Test Chart (table 1) can be used for binding media identification. An individual test or a series of parallel tests can be used to find which type of binding medium is present in a paint sample. A strong starch test indicates whether a starch binding medium is present. A strong triglyceride test indicates whether a drying oil or lard binding medium is present. (A solubility test might help differentiate between polymerized drying oil and non-polymerized lard.) A strong protein test, medium tryglyceride test, and weak phosphorus test determine the presence of an egg-based binding medium. TABLE 1 BINDING MEDIA TEST CHART If there is no previous knowledge about the character or type of binding medium used, a more systematic approach based on the Binding Media Identification Flow Chart (fig. 8) is recommended. Such an approach allows for separation of binding media, and multiple tests can be performed using a single sample.
About 5 mg of finely ground paint sample is placed into a microcentrifuge test tube. A small amount of methylene chloride (CH2Cl2) is added, and the test tube is placed in a beaker of hot water in an ultrasonic bath in a fume hood until the solvent begins to boil. Centrifugation separates the supernatant, which is tested for the presence of triglycerides (triglyceride test). The solid phase in the bottom of the test tube is boiled with distilled water for about 1 minute, mixed, and centrifuged. The resulting second supernatant is tested for the presence of blood, simple sugars, proteins, and starches. If all of the above tests are negative, a second sample should be tested for the presence of gums. Analysis of samples giving multiple positive tests should be interpreted using the Binding Media Test Chart (table 1). 9 CONCLUSIONSWorking with the proposed procedure is easy, and analytical methods for the individual tests or the whole identification system can be learned in a very short time. The procedure was originally developed for participants in “The Consolidation of Painted Ethnographic Objects,” a training course, held at the Getty Conservation Institute in June 1990. About 20 practicing conservators of ethnographic art worked with the system for 2 days. Regardless of their previous laboratory experience, all participants mastered identification procedures and were able to identify binding media types in test samples, painted facsimiles, and selected authentic ethnographic objects. The major advantage of the procedure is the fact that the majority of reagents, tools, and lab ware is readily available from laboratory supply houses (table 2); a kit can be assembled quickly and easily. A systematic instrumental approach to the identification of binding media has been published recently (Erhardt et al. 1988). The approach outlined requires the availability of infrared, gas chromatographic, and high-pressure liquid chromatographic instrumentation. While these instruments are frequently not available to a museum conservator, they do provide advantages such as ease of documentation and a somewhat smaller sample requirement. They may also provide information that is not available by our less sophisticated procedure, such as the possible identification of specific proteins, gums, or oils. Like any analytical method, our procedure is not universally applicable. Certain pigments interfere with some of the tests. Insufficient sample may be available to satisfy the requirements for some or all of the tests, or the analyst may require information concerning the presence of specific binders or drying oils. In such instances the reader is referred to the literature (Erhardt et al. 1988; Masschelein-Kleiner 1986; Mills and White 1987). Our tests were designed to provide quick answers to binding media questions at low cost to a conservation laboratory. As such, they cannot provide answers to all binding media questions or solve complex binding media “puzzles.” They can play a screening role in these complex cases, and the test results can be used to guide further investigation. In such cases, it is necessary to apply advanced instrumental techniques, and there are instances when even the most sophisticated analytical methods cannot give straightforward answers. Work is in progress to improve the analytical performance of the procedures, and we are constantly IMPORTANT NOTE: When working with the binding media tests, it is necessary to become acquainted with material safety data sheets for all reagents and practice safe and responsible laboratory procedures. ACKNOWLEDGEMENTSWe express our thanks to Frank Preusser, Neville Agnew, and Marta de la Torre of the Getty Conservation Institute for their support of the project; to Sue Walston of the Australian National Museum for numerous discussions and friendly advice; and to our colleagues Cecily Druzik and Karen Cole for their computer graphics work on text figures. REFERENCESBarham, D., and P.Trinder. 1972. An improved color reagent for the determination of blood glucose by the oxadase system. Analyst97:142. Bradford, M. M.1976. A refined and sensitive method for the quantitation of microgram quantities of protein using the principle of protein-dye binding. Analytical Biochemistry72:248. Erhardt, D., W.Hopwood, M.Baker, and D.von Endt. 1988. A systematic approach to the instrumental analysis of material finishes and binding media, AIC preprints, 16th Annual Meeting, American Institute for Conservation, Washington D.C. 67–84. Feigl, F.1966. Spot tests in organic analysis, 7th ed.Amsterdam, New York: Elsevier Publishing Co. Fiske, C. H., and Y.Subbarow. 1925. The colorimetric determination of phosphorus. Journal of Biological Chemistry66:375. Hultman, E.1959. Rapid specific method for determination of aldosaccharides in body fluids. Nature183:108. Hyvarinen, A., and E.Nikkila. 1962. Specific determination of blood glucose with o-toluidine. Clinica Chimica Acta7:140. Masschelein-Kleiner, L.1986. Scientific examination of easel paintings. PACT, Journal of the European Study Group on Physical, Chemical and Mathematical Techniques Applied to Archaeology13:186–89. McGowan, M. W., et al. 1983. A peroxidase-coupled method for the colorimetric determination of serum triglycerides. Clinical Chemistry29:538. Mills, J. S., and R.White. 1987. The organic chemistry of museum objects. London: Butterworths. Saferstein, R., ed.1982. Forensic science handbook. Englewood Cliffs, N.J.: Prentice-Hall. 272–76. Schramm, H. P., and B.Hering. 1988. Historische malmaterialien und ihre identifizierung. Graz, Austria: Akademische Druck-u. Verlagsanstalt. 192–10. Vogel. A.1989. Textbook of quantitative chemical analysis, 5th ed.New York: Longman and John Wiley and Sons. 387. AUTHOR INFORMATIONDUSAN C. STULIK graduated from Charles University, Prague, Czechoslovakia, with B.S. and M.S. degrees in chemistry. He obtained a Ph.D. degree in physics from Czechoslovakia Academy of Sciences. He is deputy director of the Scientific Research Program and head of the Analytical Section at the Getty Conservation Institute. His current research is in the application of modern scientific methods in conservation science. Address: Getty Conservation Institute, 4503 Glencoe Ave., Marina del Rey, Calif. 90292. HENRY A. FLORSHEIM received his B.S. and M.S. degrees in organic chemistry from the University of California at Los Angeles. His research interests involve the detection and identification of organic binders in paint layers. Address: Getty Conservation Institute, 4503 Glencoe Ave., Marina del Rey, Calif. 90292.
 Section Index Section Index |