THE EFFECTS OF SOLUTION APPLICATION ON SOME MECHANICAL AND PHYSICAL PROPERTIES OF THERMOPLASTIC AMORPHOUS POLYMERS USED IN CONSERVATION: Poly(vinyl acetate)sERIC F. HANSEN, MICHELE R. DERRICK, MICHAEL R. SCHILLING, & RAPHAEL GARCIA
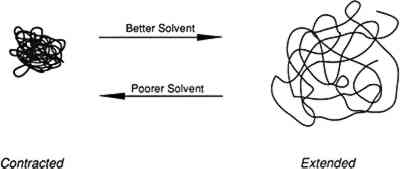
1 INTRODUCTIONWHEN SELECTING a thermoplastic polymer for a particular use as a coating, consolidant or adhesive, conservators consider the chemical stability and physical properties of the resin. Some polymers, such as poly(vinyl acetate)s (PVAC) and Acryloid B-72 (Rohm and Haas), have been shown by Feller (1978) to be relatively resistant to environmentally induced chemical modifications. These polymers have been widely used in conservation because they possess a range of physical properties suitable for the treatment of art objects. They have been designated class A materials, which by Feller's definition means that they are expected to remain in a museum environment in excess of 100 years with little comparative change in solubility. In addition to sufficient “aging” properties, physical properties that accommodate the end use in the conservation of an object are also a concern. For example, if a hard coating is required, Acryloid B-72 is a more suitable choice than the softer poly(vinyl acetate) AYAA; but the latter resin is preferred when a more flexible adhesive joint is needed.1 However, another important consideration in selecting a polymer with suitable properties that has received little attention in conservation research, is the influence of the application procedure on the properties of the polymer. When thermoplastic polymers are applied from solution, the nature of the solvent affects the conformation of the polymer in solution, which may result in an effect upon the ultimate mechanical properties, physical properties, and aging characteristics of the dry polymer film. This paper presents a short overview of the theoretical aspects that govern the ultimate physical properties of thermoplastic amorphous polymers applied from solution. The effect of solvent choice on the tensile properties, FTIR spectra, glass transition temperature (Tg) and the solvent retention of poly(vinyl acetate) cast as films is reported. Finally, suggestions are given for optimizing the solvent 1.1 THEORETICAL AND EXPERIMENTAL BACKGROUNDThe conformation of a polymer chain in solution may depend upon the solvent. One of the reasons for conformational differences is the thermodynamic “quality.” When a polymer is in a solution where the solvent has a greater attraction for segments of the polymer than the polymer segments have for each other, the chain length is extended (fig. 1). Conversely, contraction will occur when a polymer is in a solvent in which the solvent-polymer attraction is less than the polymer-polymer attraction. Solvents for a particular polymer have been denoted “good” if the chain extension is promoted and “poor” if the chain extension is contracted. Good solvents dissolve a larger amount of polymer; if the solvent is too poor, the polymer will not dissolve (Daniels and Albertyh 1975). The basis for studying solution properties of polymers is that the effects from certain conformations of the polymer chain will persist in the film deposited from such solutions, with resulting variations in physical properties of the polymeric film. However, the solvent “type” describes only the thermodynamic quality of the solution and is not necessarily connected with “good” or “bad” properties of the resultant polymer film for a specific use in the conservation of an object.
Some physical properties of many polymers have been shown to be dependent upon the solvent or solvent mixtures chosen for the solution application. This effect has been demonstrated with ethyl cellulose films on the tensile properties and the permeability to nitrogen and oxygen (Tess 1985) and on the birefringence, brittle-point temperature, and density (Haas et al. 1965). Similarly, for poly(methyl methacrylate) the surface properties of cast films (Schreiber and Croucher 1980), the hardness (Carre et al. 1980), and the shear strength (Briscoe and Smith 1983) have been shown to differ depending upon the solvent used. The time-dependent change (aging) of some mechanical and physical properties is also affected (Kurbanaliyiyev et al. 1982; and Materovsyan et al. 1985). Olayemi and Adeyeye (1982) tested the tensile properties of PVAC films cast on clean mercury surfaces from solutions of methanol, acetone, and chloroform. They determined some mechanical properties of the films after exposure at 20�C and 65% RH and at 45�C and 65�C in dry ovens. The initial modulus, yield stress, tensile strength, and work-to-rupture were shown to be greater for films cast from a solution of acetone than for those cast from a solution of chloroform. The elongation-to-break showed the reverse trend. Methanol solutions yielded films too brittle for testing. The PVAC films cast from a chloroform solution exhibited the most constant mechanical properties over the longest time interval studied (360 hours). These properties were also dependent upon the temperature of the exposure, decreasing as the temperature increased. These results were interpreted on the basis of solvent interaction with the polymer molecules both in solution and in the first stage of film formation. (In that stage, the drying rate is controlled by solvent volatility as opposed to solvent diffusion, which controls the second stage of film formation.) The results of the present study confirm those of Olayemi and Adeyeye (1982) and address further considerations: the dependence of the tensile properties of the films on the amount of retained solvent, and whether the physical properties tend toward an equilibrium value after a longer film drying time. For many polymers used in conservation the choice of solvent deserves careful consideration The results presented here provide some answers to the following questions:
The answer to this last question depends in part upon other factors that affect the final properties of a film. These questions are addressed below in the discussion of film preparation history. |