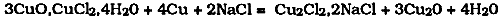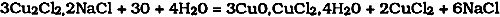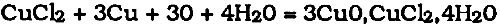BRONZE DISEASE: A REVIEW OF SOME CHEMICAL PROBLEMS AND THE ROLE OF RELATIVE HUMIDITYDAVID A. SCOTT
1 HISTORICAL INTRODUCTIONTHE CHEMICAL examination of the corrosion of copper and bronze artifacts has been the subject of study for at least 150 years. As long ago as 1826, Davy carried out an examination of a bronze helmet found in the sea near Corfu. Among the incrustations he was able to identify the ruby-red protoxide of copper (cuprous oxide: cuprite); the green rust of the carbonate (basic copper carbonate: malachite) and submuriate of copper (basic copper chloride: probably paratacamite or atacamite); crystals of metallic copper that had been redeposited; and a dirty white material identified as tin oxide. On a nail from a tomb in Ithaca that was analyzed and found to be a tin bronze with 6% of tin, Davy again found the protoxide, carbonate, and submuriate of copper, as well as tin oxide, although in this case there were no shiny crystals of redeposited copper present. The first scientific investigations of the aeruginous deposits on antiquities date from this period of the early 19th century and express the same curiosity about the nature and formation of these deposits we have today. This article summarizes the salient information published to date on the subject of cuprous chloride and bronze disease. A search of the Conservation Information Network bibliographic database (BCIN) produces about 500 references for the keywords “bronze” and “corrosion.” Many papers do not actually deal with the process of bronze corrosion but are concerned with other aspects of copper alloys and their corrosion products. All the papers dealing broadly with this topic will not be reviewed here, but some specific points will be discussed that concern the interaction of cuprous chloride with moisture; this reaction is the crucial part of the process of “bronze disease.” Bronze disease may be defined as the process of interaction of chloride-containing species within the bronze patina with moisture and air, often accompanied by corrosion of the copper alloy itself, a process which has been more or less understood for the last 100 years. The products of the reaction are light green, powdery, voluminous basic chlorides of copper, which disrupt the surface and may disfigure the object. Several corrosion processes of copper are also enhanced by visible light. Cuprous chloride, for example, is One theory concerning the origin of bronze disease attributed the problem to bacterial action. We now know that this is not the case, but the suggestion is not as silly as it sounds. Bacterial action has recently been thought to be a cause of black spots on bronzes (Madsen and Hjelm-Hansen 1979), although this theory is also unlikely to be true; a much more probable cause is the slow action of gaseous pollutants in showcases constructed of unsuitable materials that evolve sulfurous contaminants (Oddy and Meeks 1982). The best-known early attempt to deal with the problem of bronze disease is that of Berthelot, in 1895, who reported some of his proposals to account for the instability of certain bronzes. He recognized that there must be an important cyclical component to the reaction. He also realized that one of the important products of the reaction was the basic copper chloride, atacamite, which at that time was assigned the formula 3CuO,CuCl2,4H2O. The formula we know today for the copper trihydroxychlorides such as atacamite, Cu2(OH)3Cl (one of three isomeric compounds), translates closely into Berthelot's chemical terminology, which can be written as 3CuO,CuCl2,3H2O. Berthelot's explanation was a remarkable achievement for the chemistry of that time, especially since these basic copper chlorides are not well known even today. The three isomers—botallackite, atacamite and paratacamite—belong to different crystallographic groups. Botallackite is monoclinic, atacamite is orthorhombic, and paratacamite is rhombohedral. Botallackite is the least stable of the three, and its instability accounts for the rare instances in which it has been identified as a component of copper corrosion products on real objects from burial. Berthelot's explanation for the bronze disease process suggested that a small quantity of sodium chloride reacted with the atacamite and the metallic copper. A slow reaction was supposed to take place, forming a double compound of cuprous chloride and sodium chloride. The remaining portion of the copper was converted into cuprous oxide:
The double salt was oxidized by air to produce cupric chloride and atacamite:
The cupric chloride that remained in contact with the air and copper or cuprous oxide was also converted into oxychloride:
This completes the series of reactions that convert copper, oxygen, and water to cuprous oxide and atacamite in a cyclical process. Berthelot states that the constant recurrence of the process under the influence of oxygen and moisture is the cause of bronze disease. Berthelot's essential conclusion—that the recurrence is due to a cyclical reaction involving both oxygen and moisture—is indeed correct. More is known about the process today, but we still do not know all the details of the corrosion chemistry involved. The equations that Berthelot advanced and that are reproduced here are not, however, accurate descriptions of bronze disease. Although the idea of the cyclical reaction is accepted, the principal cause of instability in excavated bronze objects is due to the existence of cuprous chloride formed during corrosion processes in burial. This cuprous |