THE EFFECTS OF WASH WATER QUALITY ON THE AGING CHARACTERISTICS OF PAPERLucia C. Tang, & Norvell M. M. Jones
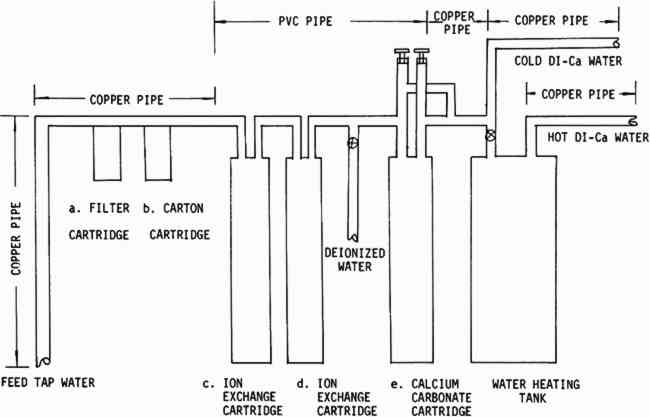
1 INTRODUCTIONWATER WASHING is a widely used treatment for conservation of paper artifacts. The conservators who use this technique believe that the water usually cleans and freshens degraded papers. After washing, old papers are often brighter and new calendered papers are commonly observed to have higher folding endurance than before washing. The water seems to remove soluble acids, degradation products and some stains and to be generally beneficial to the paper. Water washing is also often used as a final step in bleaching or organic solvent treatments. Conservators have observed that tap water is unpredictable. A dry spell, or work on the water pipes may turn the water a visible rusty color. White porcelain sinks may develop suspicious green, blue, or red-brown stains under a dripping faucet. In addition to this visible evidence of contamination by iron and copper, most urban tap water contains added chlorine, occasionally strong enough to produce odor. These three contaminants are known to be harmful to cellulose,1, 2, 3, 4 and they may be present in tap water in damaging quantities. It is not surprising that conservators have sought an alternate source of water which is uncontaminated and predictable. The logic has been that if contaminated is bad, then pure must be good, and the purer the better. Many conscientious conservators have suggested that all washing, or at least the final rinse should be done in deionized or distilled water.5 At the Library of Congress, the Preservation Office began investigating a large-scale deionized system during preparation for an extensive paper treatment program. The quantity of water necessary to wash the material was too great for existing small deionizers, and the tap water at that time was very rusty. A preliminary search suggested that little testing had been done to determine the parameters of water quality acceptable for washing valuable paper items. Iron and copper in paper are known to cause degradation when the paper is artificially aged under humid conditions;3 but the critical levels of iron and copper in the water which lead to damaging pick-up by a sheet of paper during washing are not known. High purity distilled or deionized water is recognized to be aggressive, that is very reactive. This may be an advantage or a disadvantage. Aggressive wate r should be a very effective stain remover and cleaning agent, but might also strip protective ions from the cellulose and leave it vulnerable to future degradative attack. Discussions with the representative of a major water treatment company were carried out which resulted in a system which seemed to answer the Restoration Office needs and provide some interesting possibilities for exploration. This system (Figure 1) passes the water through a 25 micron filter followed by a carbon cartridge with high adsorptive capacity for organic contaminants. The carbon also removes chlorine and chloramines. The water then passes through a weakly acidic cation exchanger column followed by a weakly basic anion exchanger. The water which has passed through both columns is relatively pure (Table VI) although it is slightly acidic due to silica and carbon dioxide which are not removed. The water then enters a third column approximately the same size as the exchanger columns, filled with calcium carbonate chips, to be neutralized and to pick up calcium to replace some of that removed in the ion exchange column. The calcium carbonate column is
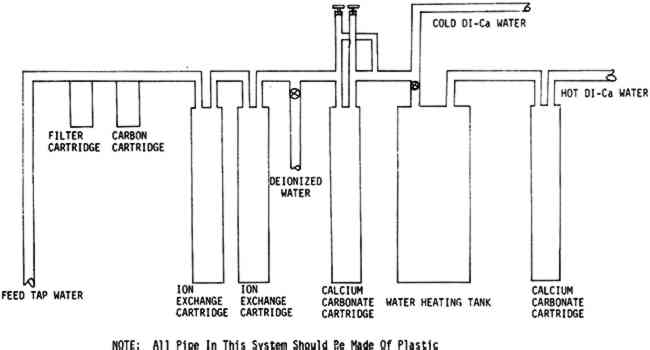
TABLE VI WATER ANALYSES FROM VARIOUS WATER SYSTEMS The design of the system was limited in part by the space available, (approximately three by six feet of floor space) and by a reluctance to replace all the existing copper plumbing until the quality of water had been fully investigated. Once the system was installed, the testing which is described in this paper was initiated to attempt to find answers to some of the questions stated above. Two modern machine-made papers were studied, as was a handmade paper from a book published in France during the late 18th Century.8 The variations in the handmade paper made it impossible to correlate the data in a meaningful way, but the trends seem to be similar to those which we observed in the modern papers. A preliminary notice of this work was published in the A. I. C. Newsletter in August, 1978. In a later issue in February, 1979, William K. Wilson of National Archives and Records Service wrote supporting our work. On the basis of test results and experience with this system, certain modifications are being made. The copper pipe will be replaced with an inert pipe to eliminate this source of possible contamination. We also plan to introduce more calcium carbonate and perhaps magnesium carbonate into the water than that obtained from the present calcium carbonate chip column. The present hot water heater is inadequate for prolonged use so more capacity for heating will be provided. One proposed revision in the system is illustrated in Figure 2.
|