AIC Preprints. May 1982. pp. 160-68
A reaction between naturally occurring glucose in egg white and amino groups on egg white proteins is described and shown to be a contributor to the highlight yellowing of albumen photographic prints. Incubation studies of plain albumen coatings and processed albumen prints reveal that yellowing is accelerated by high humidity and alkaline conditions. A specific recommendation is given against the use of alkaline buffered paper enclosures and mat boards in direct contact with albumen prints.
Approximately 80% of the photographic prints surviving from the 19th century are albumen prints. Among the various historical print processes, albumen prints are notable for the extent of yellow staining in low density (highlight) areas. The staining is so common it is often cited as a useful characteristic in identifying albumen prints. (1) The degree of highlight yellowing may range from very slight to severe. For example, the average highlight blue filter reflection density of a group of 14 albumen prints selected for display at George Eastman House was 0.39, while for a group of modern experimentally fabricated albumen prints the average was only 0.12. Otherwise similar photographic papers using gelatin instead of albumen do not show the same propensity for yellowing, although if improperly fixed any photographic material will become stained in time.
Highlight yellowing was investigated in this laboratory along with other aspects of albumen print deterioration. The yellow staining is a complex phenomenon which does not have a single, unique cause; yellowing in an actual print is a product of several mechanisms acting in concert, and it is usually not possible to determine the contribution of the various mechanisms. Major causes of highlight yellowing in albumen prints include formation of colloidal silver or silver sulfide staining from contact with highly lignified secondary supports, photochemical degradation of the albumen layer, and yellowing of the albumen due to the Maillard, or "protein-sugar" reaction. This paper will consider the role of the Maillard reaction in detail by reviewing the existing literature and by describing incubation experiments which suggest that it does play a role in albumen print highlight yellowing. The other factors in yellowing will be considered in later publications.
The reaction between reducing sugars and amine groups of amino acids and proteins is called the Maillard reaction, after the French chemist who discovered it in 1912. It has since become recognized as an important factor in determining the color, aroma, and nutritional value of many foodstuffs. During the reaction, the condensation of a reducing group of a carbohydrate and an amino group from a protein or amino acid results ultimately in a polymeric carbonyl-amine compound of low solubility. (2) The conjugated double bonds of the products of this reaction (really a series of reactions) account for their brown or yellowish coloration.
The Maillard reaction is an important consideration in albumen prints because of the significant amount of glucose naturally present in egg white proteins. (3) The modern dried egg products industry only became possible when a practical method was found to eliminate the glucose from albumen. Among the effects of leaving the glucose in the egg white are a rapid development of a brown color, loss of solubility, and rapid development of fluorescence. (4,5) The prevention of these unwanted changes in dried egg products is accomplished commercially by subjecting the egg white to a process of bacterial fermentation prior to drying. (6) The fermentation process significantly lowers the amount of glucose and allows for a much more stable product.
In order to evaluate the practical significance of the Maillard reaction in albumen print highlight yellowing, coatings were made with both fresh and fermented albumen. Fresh egg white was beaten in a household blender for 1-1 / 2 minutes on high speed, allowed to settle back to liquid form, then incubated in loosely covered containers at 30°C Fermentation of egg white in this manner was described in an article by Stuart and Goresline (7) and in several places in the 19th-century literature on albumen paper manufacturing. (8,9) Unless speeded up by inoculation with appropriate bacteria, the process takes about 10 days; a foamy white layer rises to the surface of the solution while a finely textured precipitate sinks to the bottom. Both these are removed prior to coating. The pH of the albumen falls during the process from approximately 9.0 to 6.0-6.5. completion.
The presence of glucose was monitored with ClinistixTM a semi-quantitative but reliable qualitative detector of glucose. Prior to fermentation the albumen tested strongly positive for glucose and was not at all fluorescent when illuminated with a hand-held short wave UV source. After 6 days the pH was 8.5, and the albumen still tested strongly positive for glucose. At this point samples were coated with the glucose-containing egg white and designated as "fresh" samples. The paper stock used in these experiments was a 60 gram rag photobase, without baryta coating. After 10 days, the albumen tested negative for glucose and the pH was 6.42. The albumen was not fluorescent. Samples were now coated with the fermented albumen solution.
The fresh and fermented unsensitized albumen paper samples as described above were incubated at 50°C and four different relative humidities—30, 50, 75, and 95%. The results after 30 days incubation are summarized in Table 1, which presents blue filter density changes (blue filter densities provide the most sensitive indicator of yellowing of the albumen).
Table I.
Yellowing of Unsensitized Albumen Coatings At Various RH Levels
| INCUB RH | 30% | 50% | 75% | 95% |
| FRESH | .02 | .04 | .06 | .05 |
| FERMENTED | .02 | .02 | .02 | .03 |
The results show that the fermented samples with the natural glucose removed yellowed considerably less than the samples coated from the same batch of albumen prior to glucose removal. The fresh, glucose-containing samples displayed a vivid bluish-white fluorescence which increased in proportion to the RH during incubation and the amount of yellowing present The fermented samples did not fluoresce nearly as much as the fresh samples. The existence of a maximum of yellowing at the 75% level for fresh albumen samples in this experiment does coincide with published accounts of the effect of ambient moisture on the course of the Mallard reaction. Hannan and Lea studied an amino acid-glucose Model system and reported a maximum of color development at 60% RH. (10) Thus the effect of moisture on the yellowing of albumen prints will be different for glucose-containing and non glucose-containing prints. Albumen prints made with fresh albumen can be expected to yellow more in non-image areas than comparably processed prints made with fermented, glucose-free albumen.
In addition to the coatings of plain, unsensitized albumen described above, actual albumen prints were created using fresh and fermented egg white. These were incubated together at various RH's and temperatures to compare the effects of desugarization on highlight yellowing Both sets of samples received the same sensitization, exposure, and processing. Table 2 presents the average red and blue filter reflection density changes (compared to unincubated controls) from 30 days of incubation at 50°C and 95% RH. The data are averages of eight duplicates. It is apparent from the data that the fresh, glucose-containing albumen samples yellowed more in non-image areas during incubation than did the desugared, fermented albumen samples. The extra blue filter density gain in a minimum density area and the lower levels of blue filter density loss in highlight and shadow (high density) areas in the fresh samples demonstrate the increased yellowing. This agreed quite well with the results of incubation of unsensitized albumen coatings of fresh and fermented egg white, where the fresh samples also yellowed more than the fermented.
| FRESH | FERMENTED | |||
| RED | BLUE | - RED | BLUE | |
| D-MIN | 0 | +.05 | 0 | +.02 |
| HIGHLIGHT AREA | -.03 | +.02 | -.04 | -.04 |
| HIGH DENSITY | -.23 | -.03 | -.24 | -.11 |
The experiments with albumen fermentation show that the amount of glucose present can affect the degree of highlight yellowing of albumen prints. The literature on the Maillard reaction describes three important influences on the course of the reaction: temperature, (11) ambient moisture, (10) 3 pH. (12) These characteristics have implications for the conservation treatment and storage of albumen prints. The first is temperature; the Mallard reaction is extremely thermally sensitive. The reaction may be initiated by a period of high heating, then go to at room temperature, with an overall rate much faster than if the sample had never been initially heated. (11)
In dry mounting of photographs the temperature required for all commonly used mounting tissues is above 75°C. Although it has not been verified experimentally, it is quite likely that even such a brief period of heating would accelerate Mallard yellowing of albumen prints and should be avoided. This would therefore exclude from conservation practice for albumen prints all heat-set mounting techniques or other conservation measures involving heat because of the danger of accelerating yellowing and unwanted physical changes in the albumen. The role of moisture in accelerating yellowing was shown in the experiments described in Section 4; as ambient moisture increased, so did the level of yellowing in both plain albumen coatings and processed albumen prints.
The role of pH in albumen print yellowing is an important one, with consequences for both storage and treatment of albumen photographs.
According to the literature the reaction is retarded below pH 6 and accelerated by alkaline conditions. (12) This assumption was tested in incubation experiments of two kinds, one assessing the broad effects of pH, and the other focusing specifically on how contact with alkaline materials in storage at high humidity affects the yellowing process. In both cases yellowing proved to be stimulated by alkaline conditions.
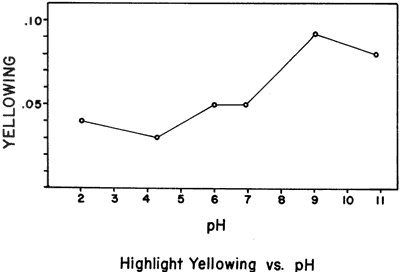
In order to assess the broad effects of pH differences on albumen print highlight yellowing, normally processed, non-fermented albumen print samples (13) were soaked for 18 hours in solutions of various re-agents, then blotted and dried without rinsing. The reagents and their solution pH's were: 1) 0.1 N H2S04: pH 2.0, 2) 0.5 M HKC8H4O4: pH 4.3, 3) distilled water: pH ca. 6, 4) 0.25 M KH2PO4 + 0.25 M NaH2PO4: pH 6.9, 5) 0.1 M Na2B4O7 . 10 H20: pH 9.1, and 6) 0.1 N NaOH: pH 10.9. After soaking, the samples were incubated at 50°C, 95% RH for 60 days. The amount of highlight yellowing (expressed as blue filter density gain in a non-image area) is plotted against the original solution pH in Figure 1. The data points are averages of two duplicates. In this experiment alkaline conditions clearly increased the amount of yellowing produced upon incubation.
Figure 1
Since alkaline buffered papers and boards are so widely used for storage enclosures and mats for albumen prints, an experiment was undertaken to evaluate whether contact with alkaline papers during incubation would accelerate the yellowing process. The basic approach used was that of the Photographic Activity Test described in ANSI Standard PH 1.53-1978, "Filing Enclosures for Processed Photographic Films, Plates and Papers". In this type of incubation test the enclosure material under study is incubated in contact with a sample photograph at 50°C and 86% RH. Control samples are incubated in contact with filter paper, and these serve as the basis for evaluation of the enclosure material being tested. In one trial, normally-processed, non-fermented albumen print samples were incubated at 86% RH, 50°C for 150 days in contact with some common enclosure materials. Table 3 gives the type of material, together with the yellowing (blue filter density gain in a non-image area) produced after 150 days incubation. The data are average changes from 4 duplicate samples, except for filter paper where 16 duplicate samples are averaged.
The results of the Photographic Activity Test presented in Table 3 suggest that carbonate buffered papers such as Permalife may lead to increased yellowing under humid conditions if kept in close contact with albumen prints. Two repetitions of this type of incubation test gave similar results and supported the conclusion that alkaline conditions promote non-fermented albumen print samples.
For this reason alkaline buffered paper enclosures and mat boards should not be used in direct contact with albumen prints. Several other points also should be noted: 1) high humidity is required in order for the alkalinity of the enclosure to migrate to the print, 2) direct contact between the enclosure material and the print is necessary for the print to be adversely affected, and 3) the amount of additional yellowing produced in these experiments by alkaline conditions is not so severe as to mandate hasty changes in storage enclosures. Rather, this evidence suggests that an alternative to alkaline mat boards and enclosures is needed for albumen prints. These alternative materials—perhaps high alpha-cellulose, non-buffered papers—should be thoroughly tested in incubation studies with albumen prints, then gradually integrated into collections to replace existing enclosures and house new acquisitions.
The incubation experiments described above indicate that the amount of glucose contained in albumen at the time it is coated with have an effect on the extent and nature of subsequent deterioration. Bacterial fermentation was part of the manufacturing process for much of the albumen paper made in the 19th century. (14) A survey of 19th-century literature on albumen paper manufacturing indicates that most early prints (from the era 1850-1880) were coated with fresh albumen, while most later prints (from the era 1880-1900) were coated with fermented albumen. Prior to 1860 the majority of albumen paper consumed was coated by individual photographers but after that time the preparation of albumen paper tended to become centralized in factories. For a number of reasons the industry gradually became geographically centralized in Dresden, Germany. Fermentation appears to have been a consistent practice in the Dresden operations from the late 1870's on.
Outside Dresden, and particularly in the United States, fresh albumen seems to have been the norm. There were many references in the literature and advertising of the period to the mild odor of German albumen paper. Despite much discussion of the merits of fermentation, it continued to be standard practice in Dresden and in at least a few other places as long as albumen paper continued to be manufactured. The importance of Dresden as a manufacturing center grew tremendously during the 1870's and 1880's, and so did the numbers of prints worldwide made with fermented albumen. By the 1890's American photographers were using only 20-30% domestically coated paper, with the rest coming principally from Dresden. The high point of American manufacturing of albumen paper came in the late 1860's and 1870's, and most if not all of the product was coated with fresh albumen.
The implications of the history of albumen paper manufacturing are that earlier prints will have more Maillard yellowing as a group than later prints, the majority of which were coated with desugared egg white . This hypothesis was born out in the examination of a large number of family photograph albums from the 1865-1895 period. In such albums are many vignetted portraits with considerable non-image area in which to observe the yellowing. In the large majority of observations the yellowing of earlier, "carte de visite" albumen prints dating from the 1860's and early 1870's was found to be more severe than that encountered in later 1880's and 1890's studio work. The additional yellowing is more than the extra age of the prints would allow for, and does suggest that the Maillard reaction may be part of the reason for the extra yellowing observed in the early prints.
The reaction between naturally occurring glucose in egg white and amino groups on the egg proteins has been shown to be a contributing factor to the highlight yellowing of albumen photographic prints. However, there are other important factors leading to highlight yellowing, and it is not possible to determine the relative amount of Maillard yellowing in any given print. High temperature, high relative humidity, and alkaline conditions all promote the Maillard reaction, so that proper storage of albumen prints will avoid these conditions. Recommended temperature and humidity conditions for the storage of albumen prints are:.
| TEMPERATURE: | LESS THAN 18°C |
| RH: | 30-40% |
Provisional recommendations for filing enclosure materials for albumen prints are uncoated polyester and cellulose triacetate.
1. Rempel, S., "The Care of Black and White Photographic Collections Identification of Processes", Technical Bulletin #6, Canadian Conservation Institute, Ottawa (1979), p. 21.
2. Kato, Y., Watanabe, K., and Sato, Y., Effect of the Maillard Reaction on the Attributes of Egg White Proteins, Agricultural and biological Chemistry, 42 (1978), pp. 2233-2237.
3. Powrie, W., "Chemistry of Eggs and Egg Products", in Egg Science and Technology, 2nd ed., AVI Publishing, Westport Connecticut (1977), pp. 65-91.
4. Hill, W. M., and Sebring, M., "Desugarization," in Egg Science and Technology, 2nd ed., AVI Publishing, Westport, Connecticut (1977), pp. 187-196.
5. Stewart, G., and Kline, R., Dried Egg Albumen. I. Solubility and Color Degradation, Proceedings of the Institute of Food Technology (1941), pp. 48-56.
6. Berquist, D. H., "Egg Dehydration", in Egg Science and Technology, 2nd ed., AVI Publishing, Westport Connecticut (1977), pp. 197-229.
7. Stuart, L., and Goresline, H., Bacteriological Studies on the "Natural" Fermentation Process of Preparing Egg White for Drying, J. Bacteriology, 44 (1942), pp. 541-549.
8. Miethe, A., Lehrbuch der Praktischen Photographie, Wilhelm Knapp, Halle (1902), pp. 262-284.
9. Eder, J. M., Die Photographische Kopierverfahren mit Silbersalzen (Positiv-Prozess), Wilhelm Knapp, Halle (1898), pp. 121-122.
10. Hannan, R., and Lea, C., Studies of the Reaction Between Proteins and Reducing Sugars in the "Dry" State. VI. the Reactivity of the Terminal Amino Groups of Lysine in Model Systems, Biochimica et Biophysica Acta, 9 (1952), pp. 293-305.
11. Spanyar, P., "The Importance of the Maillard Reaction in Food Chemistry", Period. Polytech. Chem. Eng., 20 (1976), pp. 371-378.
12. Bergquist, D. H., "Egg Dehydration" in Egg Science and Technology, AVI Publishing, Westport Connecticut (1977), p. 207.
13. Reilly, J., "Image Deterioration in Albumen Photographic Prints", Preprints of the 9th International Congress of the International Institute for Conservation, 1982 (In Press). Details of albumen print sample preparation are given here.
14. Reilly, J., The Albumen and Salted Paper Book: The History and Practice of Photographic Printing, 1840-1895, Light Impressions, Rochester (1980), pp. 27-46 (The discussion of manufacturing here is referenced; consult these references for the source material used).